Chapter 10: Q43P (page 537)
Suggest carbonyl compounds and reducing agents that might be used to form the following alcohols.
- heptan-1-ol
- 1-cyclohexylethanol
- 1-phenylpropan-1-ol
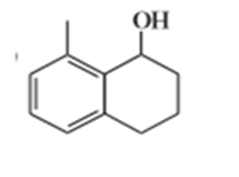
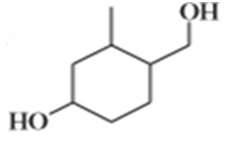
e)
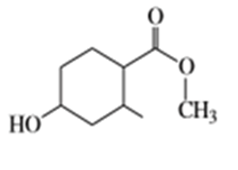
f)
Short Answer
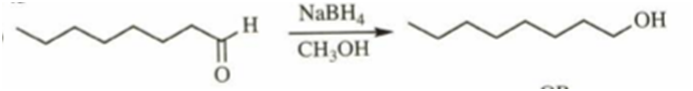
a)
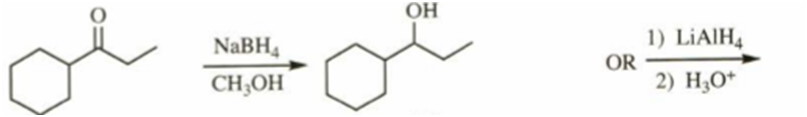
b)
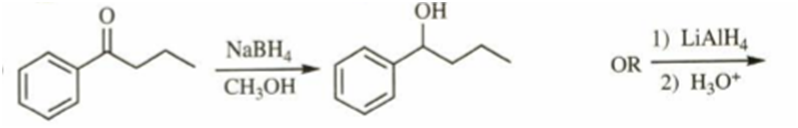
c)
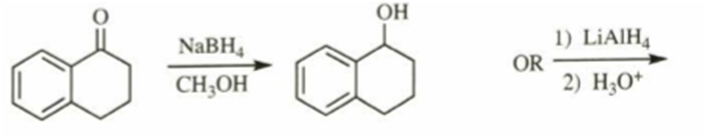
d)
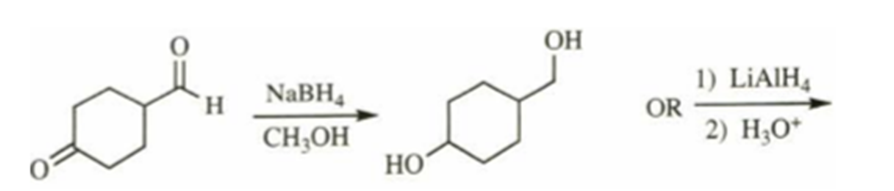
e)
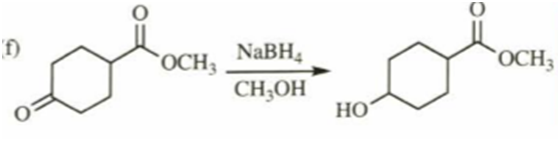
f)
Learning Materials
Features
Discover
Chapter 10: Q43P (page 537)
Suggest carbonyl compounds and reducing agents that might be used to form the following alcohols.


e)

f)

a)

b)

c)

d)

e)

f)
All the tools & learning materials you need for study success - in one app.
Get started for free
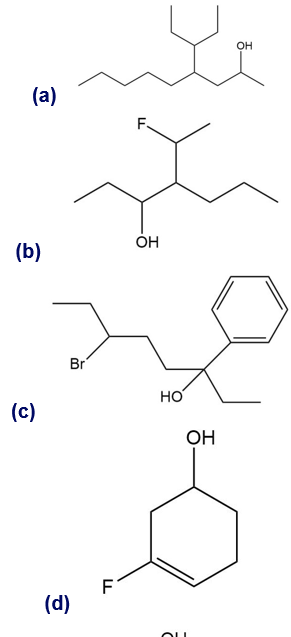
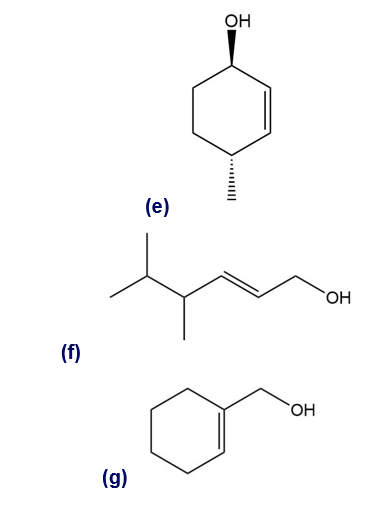
Give a systematic (IUPAC) name for each alcohol. Classify each as primary, secondary, or tertiary.
Show how you would add Grignard reagents to acid chlorides or esters to synthesize the following alcohols.
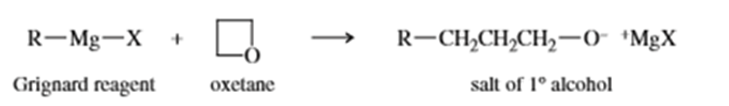
Grignard reagents react slowly with oxetane to produce primary alcohols. Propose a mechanism for this reaction, and suggest why oxetane reacts with Grignard reagents even though most ethers do not.
Vinyl alcohols are generally unstable, quickly isomerizing to carbonyl compounds. Propose mechanisms for the following isomerizations.
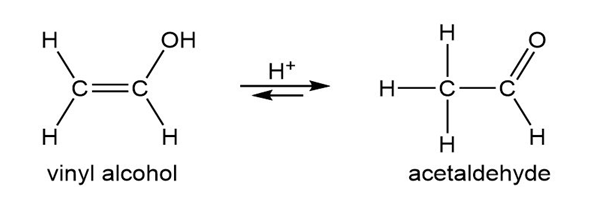
(a)
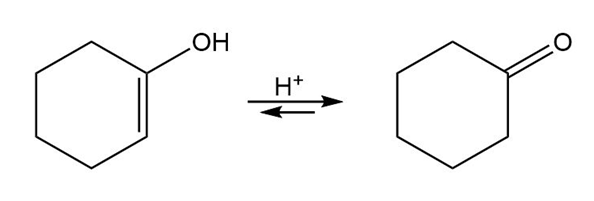
(b)
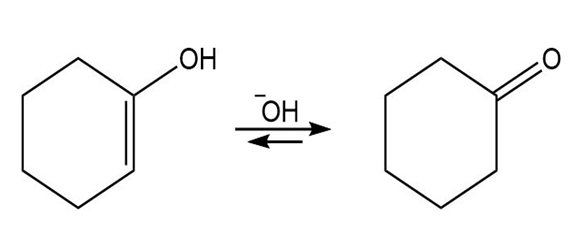
c)
Show how you would synthesize each tertiary alcohol by adding an appropriate Grignard reagent to a ketone.
(a)3-phenylhexane-3-ol (3ways)
(b) Ph3 COH
(c) 1-ethylcyclopentanol
(d) 2-cyclopentylpentan-2-ol
What do you think about this solution?
We value your feedback to improve our textbook solutions.