Chapter 10: Q46P (page 538)
Vinyl alcohols are generally unstable, quickly isomerizing to carbonyl compounds. Propose mechanisms for the following isomerizations.

(a)
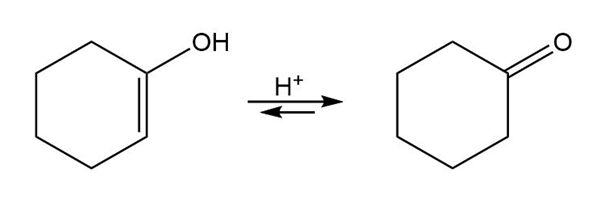
(b)

c)
Short Answer
a)
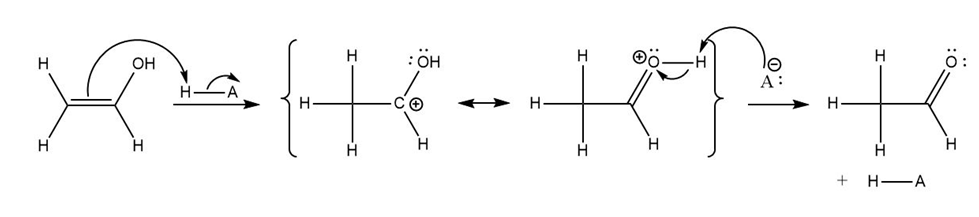
b)

c)
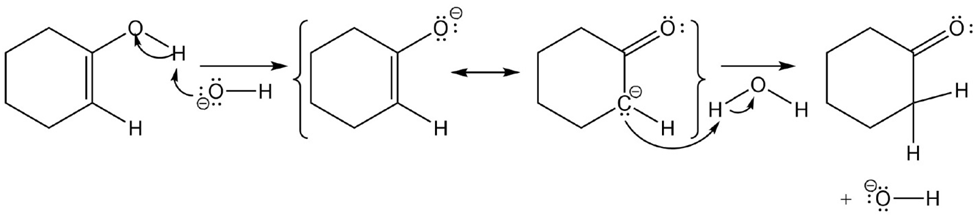
Learning Materials
Features
Discover
Chapter 10: Q46P (page 538)
Vinyl alcohols are generally unstable, quickly isomerizing to carbonyl compounds. Propose mechanisms for the following isomerizations.

(a)

(b)

c)
a)

b)

c)

All the tools & learning materials you need for study success - in one app.
Get started for free
For each synthesis, start with bromocyclohexane and predict the products. Assume that an excess of each reactant is added so that all possible reactions that can happen will happen.
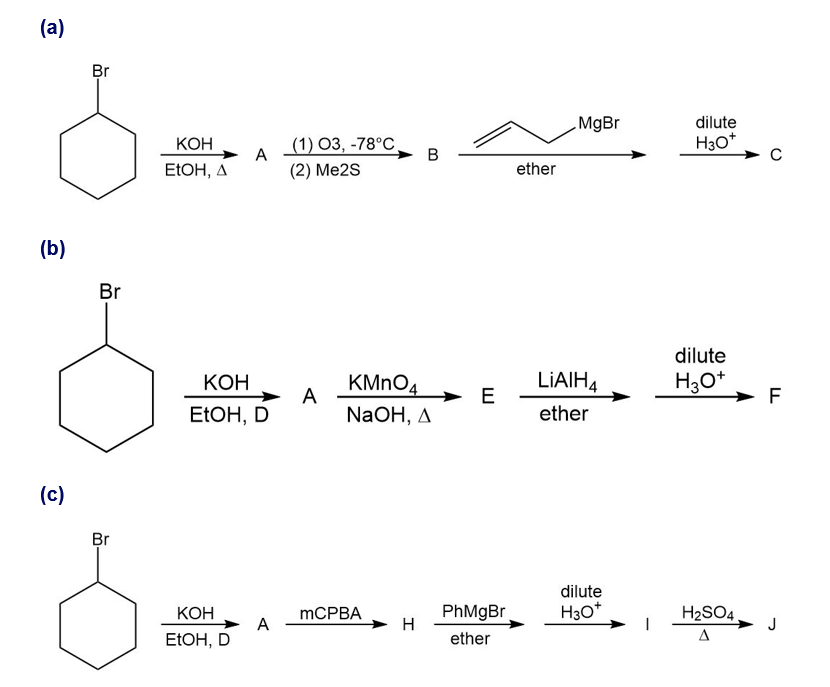
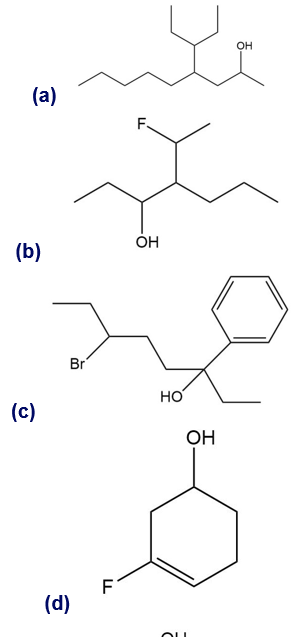
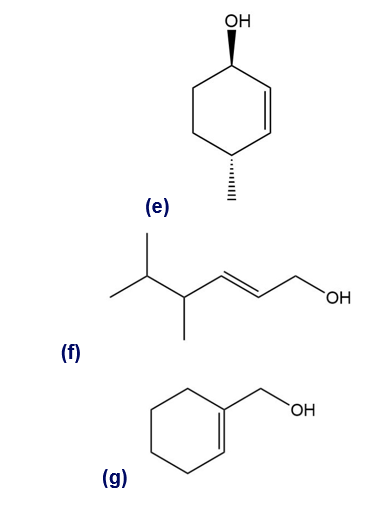
Give a systematic (IUPAC) name for each alcohol. Classify each as primary, secondary, or tertiary.
Arrange the following compounds in order of decreasing acidity.
CH3COOH CH3OH CH3CH3 CH3SO3H CH3NH2 CH3SH CH3C CH
CH
Give the IUPAC name of the following alcohols.
Authentic skunk spray has become valuable for use in scent-masking products. Show how you would synthesize the two major components of skunk spray (3-methylbutane-1-thiol and but-2-ene-1-thiol) from any of the readily available butenes or from buta-1,3-diene.
What do you think about this solution?
We value your feedback to improve our textbook solutions.