Chapter 10: Q58P (page 540)
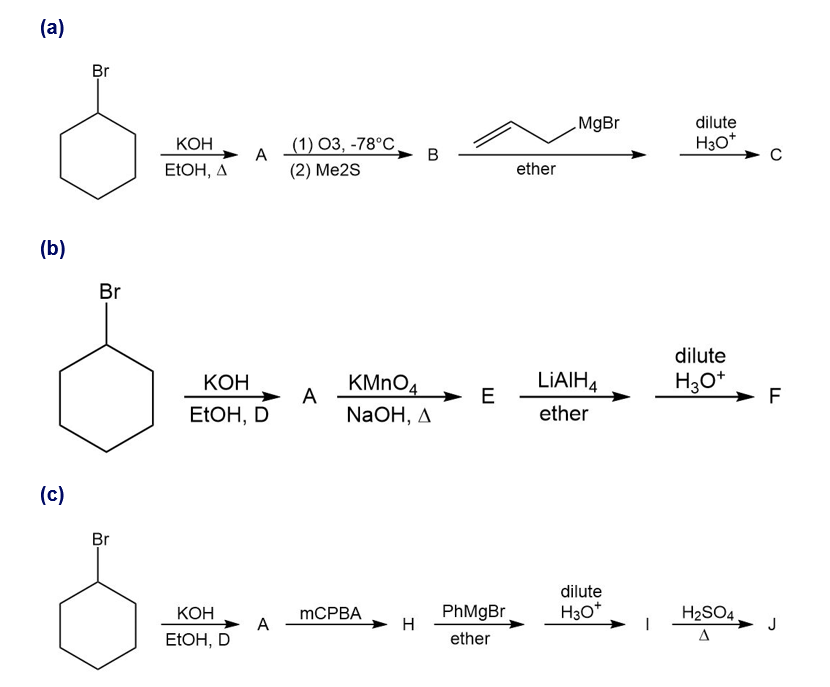
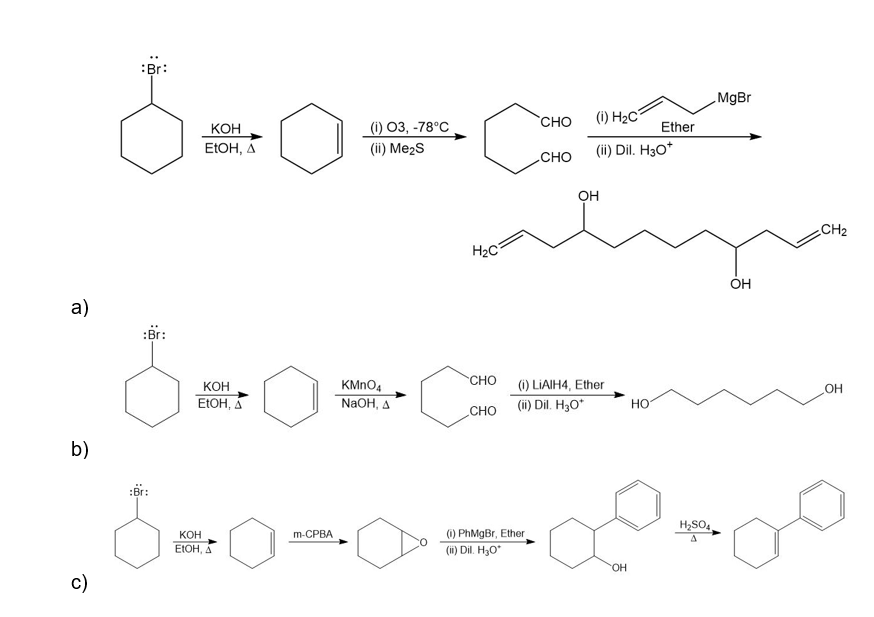
For each synthesis, start with bromocyclohexane and predict the products. Assume that an excess of each reactant is added so that all possible reactions that can happen will happen.
Short Answer
Learning Materials
Features
Discover
Chapter 10: Q58P (page 540)
For each synthesis, start with bromocyclohexane and predict the products. Assume that an excess of each reactant is added so that all possible reactions that can happen will happen.


All the tools & learning materials you need for study success - in one app.
Get started for free
Suggest carbonyl compounds and reducing agents that might be used to form the following alcohols.
e)
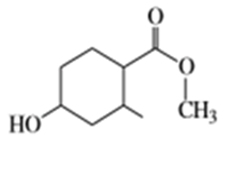
f)
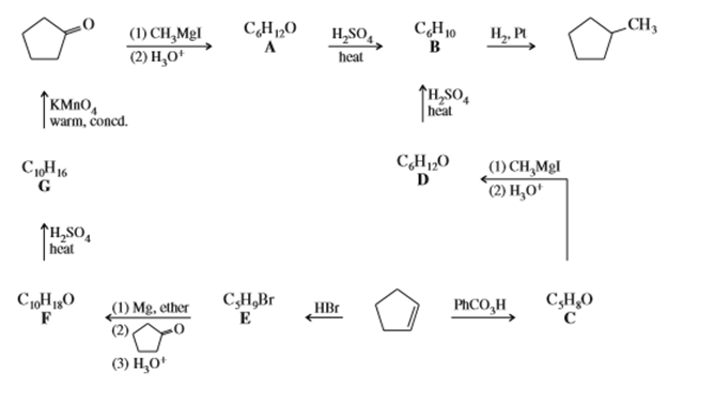
Determine the structures of compounds A through G, including stereochemistry where appropriate.
Give a systematic (IUPAC) name for each diol.
a.
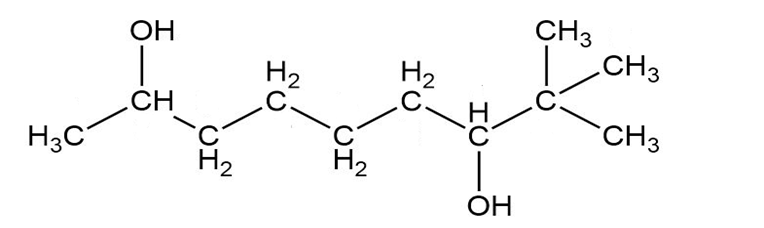
b.
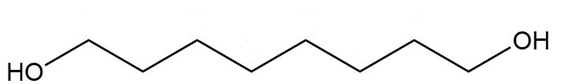
c.
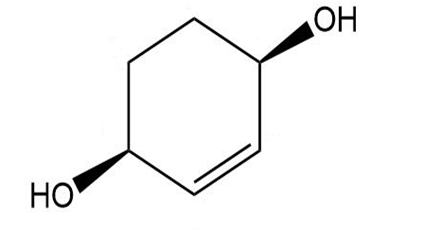
d.
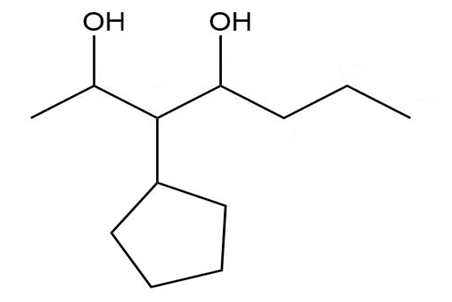
e.
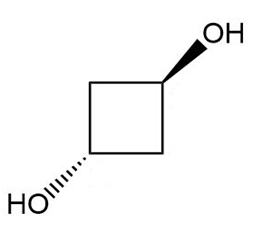
Give the IUPAC name of the following alcohols.
Predict which member of each pair will be more soluble in water. Explain the reasons for your answers.
(a) hexan-1-ol or cyclohexanol
(b) heptan-1-ol or 4-methylphenol
(c)3-ethylhexan-3-ol or octan-2-ol
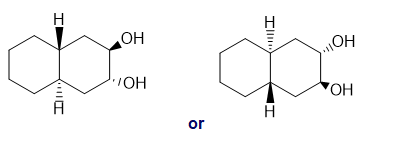
(d)hexan-2-ol or cyclooctane-1,4-diol
(e)
What do you think about this solution?
We value your feedback to improve our textbook solutions.