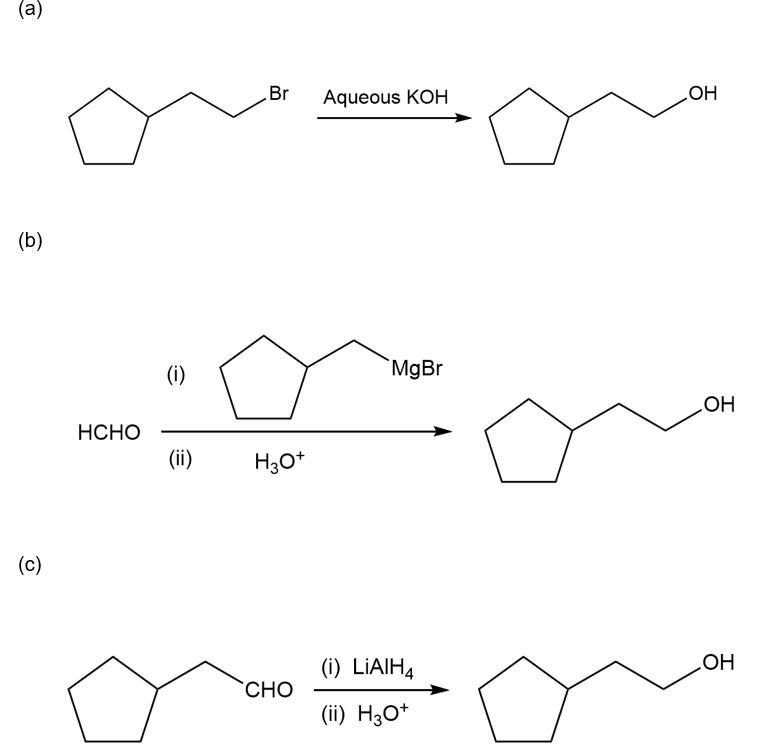
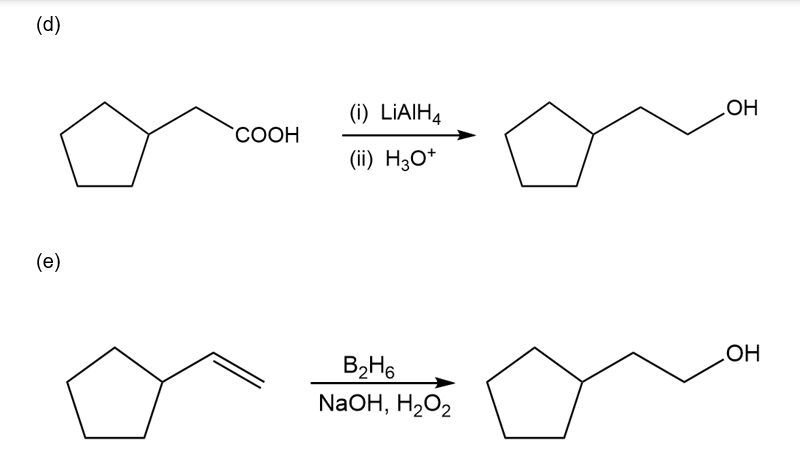
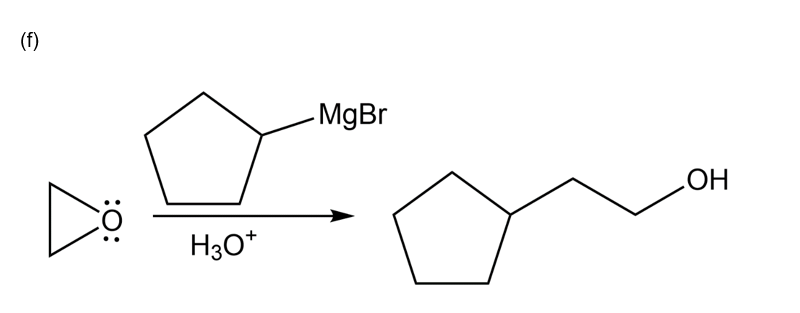
Chapter 10: Q57P (page 540)
Show how this 1° alcohol can be made from the following:
(a) a 1° alkyl bromide
(b) formaldehyde
(c) a 7-carbon aldehyde
(d) a carboxylic acid
(e) an alkene
(f) ethylene oxide
Short Answer
Learning Materials
Features
Discover
Chapter 10: Q57P (page 540)
Show how this 1° alcohol can be made from the following:
(a) a 1° alkyl bromide
(b) formaldehyde
(c) a 7-carbon aldehyde
(d) a carboxylic acid
(e) an alkene
(f) ethylene oxide



All the tools & learning materials you need for study success - in one app.
Get started for free
Complete the following acid-base reactions. In each case, indicate whether the equilibrium favors the reactants or the products, and explain your reasoning.
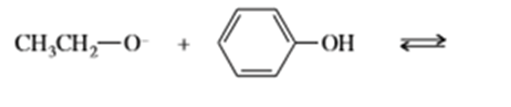
a)
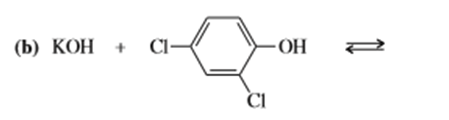
b)
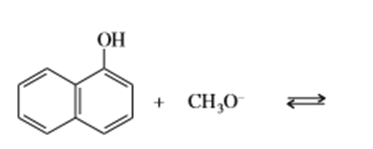
c)
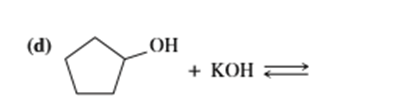
d)
e) (CH3)3 C-O- + CH3CH2OH⇄
f)(CH3)3 C-O- + H2O ⇄
g) KOH+ CH3CH2OH ⇄
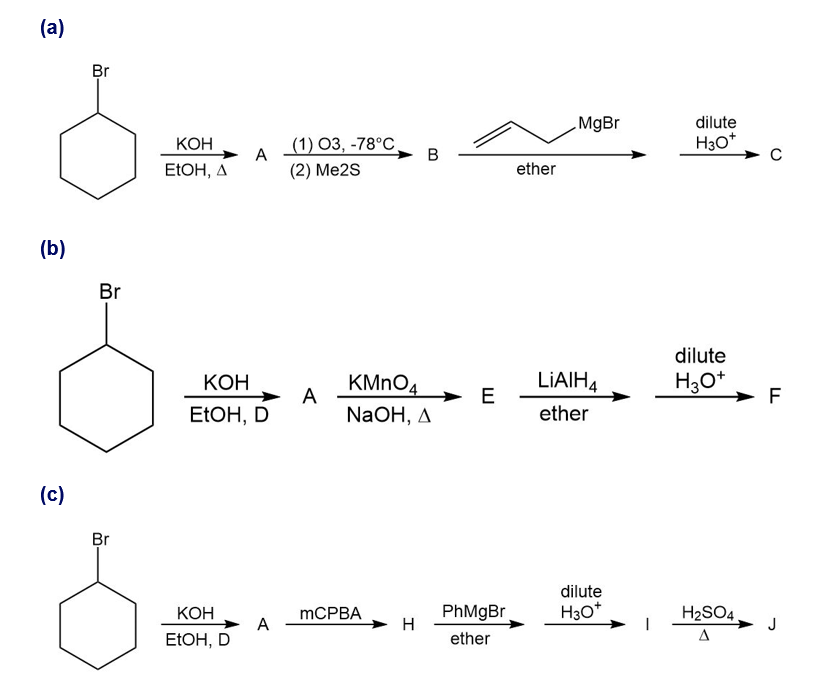
For each synthesis, start with bromocyclohexane and predict the products. Assume that an excess of each reactant is added so that all possible reactions that can happen will happen.
Question: Show how you would synthesize the following primary alcohols by adding an appropriate Grignard reagent to formaldehyde.
a.
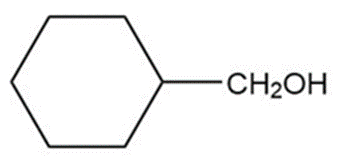
b.
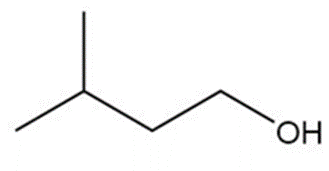
c.
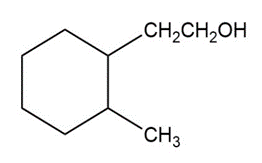
Show how you would synthesize the following alcohols by adding Grignard reagents to ethylene oxide.
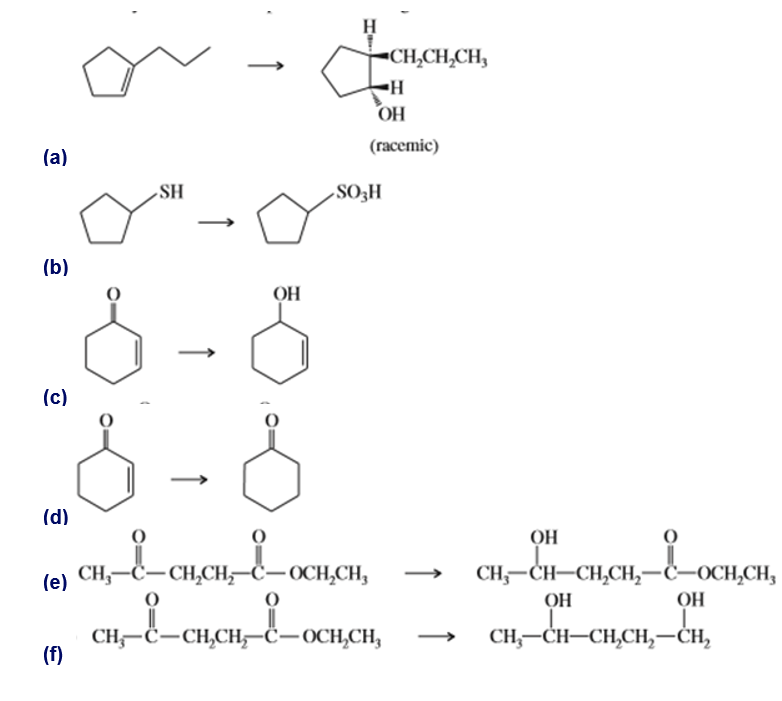
Show how you would accomplish the following transformations. You may use any additional reagents you need.
What do you think about this solution?
We value your feedback to improve our textbook solutions.