Chapter 10: Q56P (page 540)
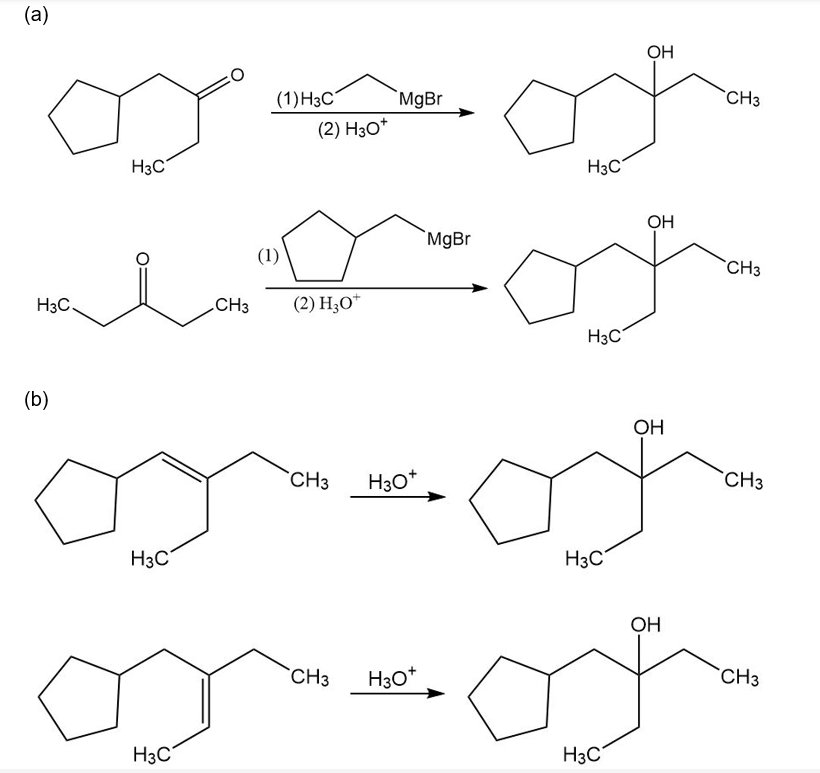
Often, compounds can be synthesized by more than one method. Show how this 3° alcohol can be made from the following:
(a) two different ketones
(b) two different alkenes
(c) an ester
(d) a 3° alkyl bromide
Short Answer
Learning Materials
Features
Discover
Chapter 10: Q56P (page 540)
Often, compounds can be synthesized by more than one method. Show how this 3° alcohol can be made from the following:
(a) two different ketones
(b) two different alkenes
(c) an ester
(d) a 3° alkyl bromide


All the tools & learning materials you need for study success - in one app.
Get started for free
Devise a synthesis for each compound, starting with methylenecyclohexane and any other reagents you need.
(a) 1-methylcyclohexanol (b) cyclohexylmethanol
(c) 1-(hydroxymethyl)cyclohexanol (d) trans-2-methylcyclohexanol
(e) 2-chloro-1-methylcyclohexanol (f) 1-(phenylmethyl)cyclohexanol
Show how you would synthesize the following alcohols by reducing appropriate carbonyl compounds.
a. Heptan-1-ol (b) Heptan-2-ol (c) 2-methylhexan-2-ol
b.
Authentic skunk spray has become valuable for use in scent-masking products. Show how you would synthesize the two major components of skunk spray (3-methylbutane-1-thiol and but-2-ene-1-thiol) from any of the readily available butenes or from buta-1,3-diene.
Show how you would accomplish the following transformations. You may use any additional reagents you need.
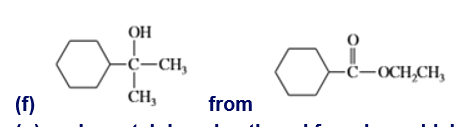
g. cyclopentylphenylmethanol from benzaldehyde
h. octan-1-ol from 1-bromohexane
The following compounds are only slightly soluble in water, but one of them is very soluble in a dilute aqueous solution of sodium hydroxide. The other is still only slightly soluble.
(a) Explain the difference in solubility of these compounds in dilute sodium hydroxide.
(b) Show how this difference might be exploited to separate a mixture of these two compounds using a separatory funnel.
What do you think about this solution?
We value your feedback to improve our textbook solutions.