Chapter 10: Q13P (page 516)
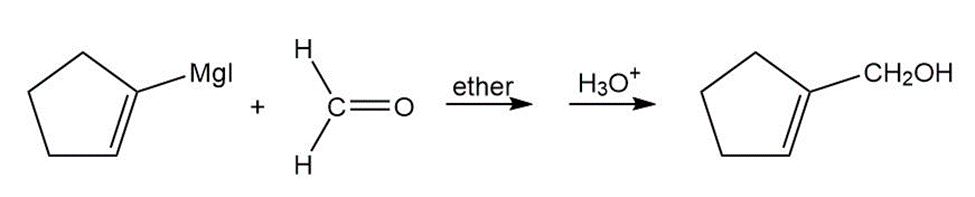
Question: Show how you would synthesize the following primary alcohols by adding an appropriate Grignard reagent to formaldehyde.
a.
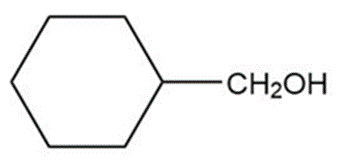
b.
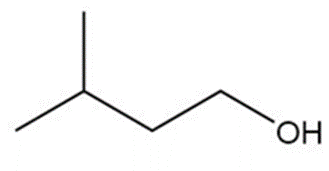
c.
Short Answer
Answer
a.
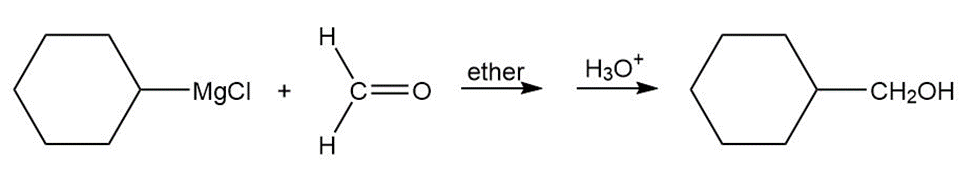
b.
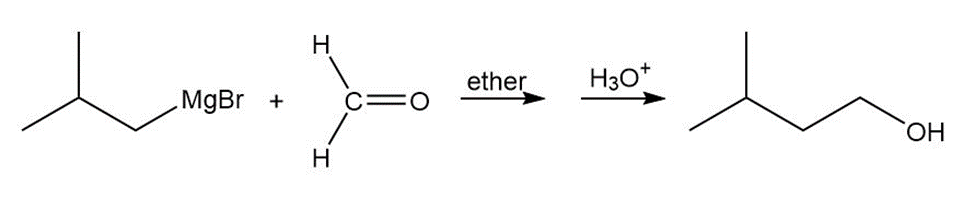
c.
Learning Materials
Features
Discover
Chapter 10: Q13P (page 516)
Question: Show how you would synthesize the following primary alcohols by adding an appropriate Grignard reagent to formaldehyde.
a.

b.

c.
Answer
a.

b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
Show how to make these deuterium-labeled compounds, using CD3MgBrand D2Oas your sources of deuterium, and any non-deuterated starting materials you wish.
(a) CH3CH(OD)CD3(b) CH3C(OH)(CD3)2 (c) CD3CH2CH2O(d) Ph(CD3)2COD
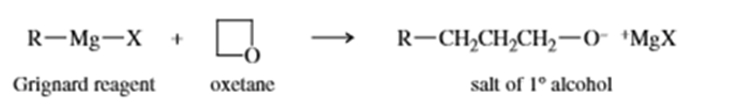
Grignard reagents react slowly with oxetane to produce primary alcohols. Propose a mechanism for this reaction, and suggest why oxetane reacts with Grignard reagents even though most ethers do not.
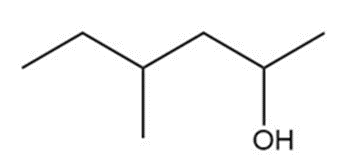
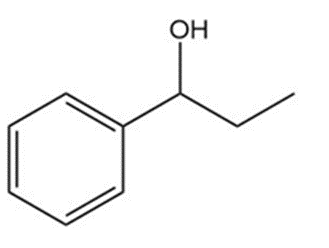
Question: Show two ways you could synthesize each of the following secondary alcohols by adding an appropriate Grignard reagent to an aldehyde.
a.
b.
c.
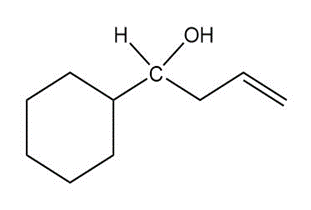
Give both the IUPAC name and the common name for each alcohol.
(a)
(b)
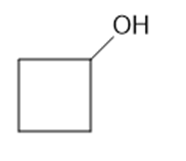
(c)
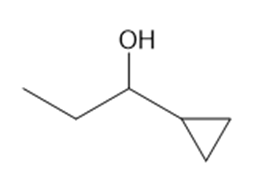
(d)
Show how you would accomplish the following transformations. You may use any additional reagents you need.
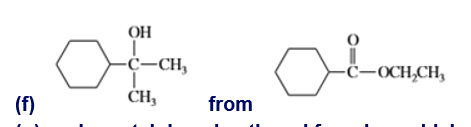
g. cyclopentylphenylmethanol from benzaldehyde
h. octan-1-ol from 1-bromohexane
What do you think about this solution?
We value your feedback to improve our textbook solutions.