Chapter 10: Q48P (page 538)
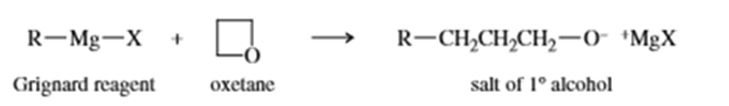
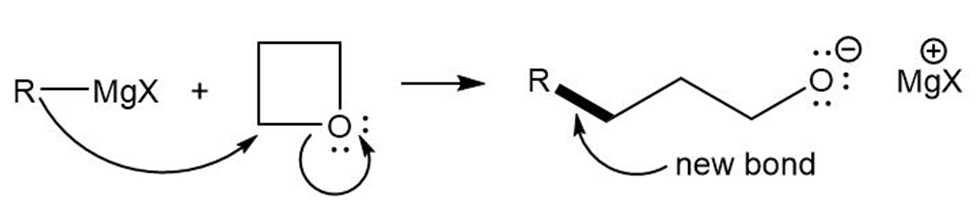
Grignard reagents react slowly with oxetane to produce primary alcohols. Propose a mechanism for this reaction, and suggest why oxetane reacts with Grignard reagents even though most ethers do not.
Short Answer
Learning Materials
Features
Discover
Chapter 10: Q48P (page 538)
Grignard reagents react slowly with oxetane to produce primary alcohols. Propose a mechanism for this reaction, and suggest why oxetane reacts with Grignard reagents even though most ethers do not.


All the tools & learning materials you need for study success - in one app.
Get started for free
Show how you would accomplish the following transformations. You may use any additional reagents you need.
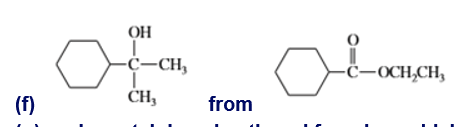
g. cyclopentylphenylmethanol from benzaldehyde
h. octan-1-ol from 1-bromohexane
Give IUPAC names for the following compounds.
a.
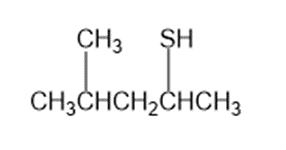
b.
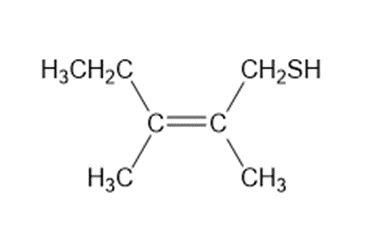
c.
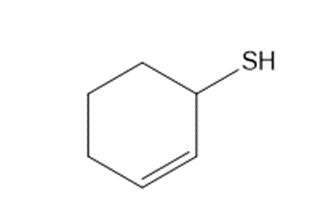
Arrange the following compounds in order of decreasing acidity.
CH3COOH CH3OH CH3CH3 CH3SO3H CH3NH2 CH3SH CH3C CH
CH
Question: Predict the products of the following reactions.
(a)
(b)
(c)
(d)
Ever since the Grignard reaction was discovered to react with ethylene oxide, chemists have experimented with other epoxides to test the limits of this reaction (which chemists call the scope of the reaction). The conclusion is that the success of the Grignard reaction is limited by the steric hindrance in the epoxide. One group published results of several Grignard reagents on one epoxide; one of their products is shown here. What Grignard reagent and what epoxide were used to make this compound? Comment on the steric requirements of the epoxide and the stereochemistry of the product.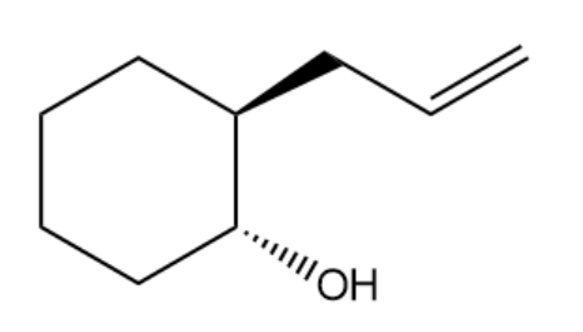
What do you think about this solution?
We value your feedback to improve our textbook solutions.