Chapter 10: Q17P (page 519)
Show how you would add Grignard reagents to acid chlorides or esters to synthesize the following alcohols.
- Ph3C-OH
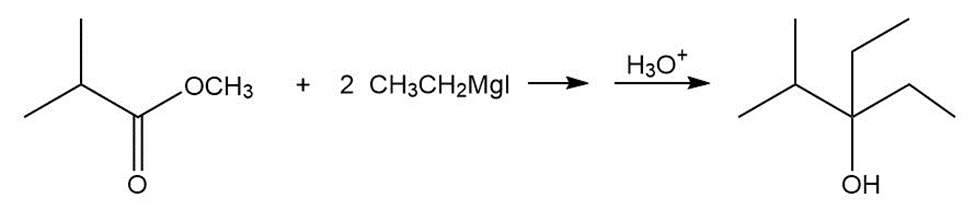
- 3-ethyl-2-methylpentan-3-ol
- dicyclohexylphenylmethanol
Short Answer
a.
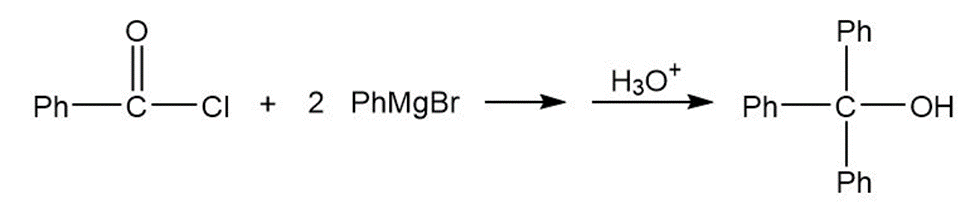
b.
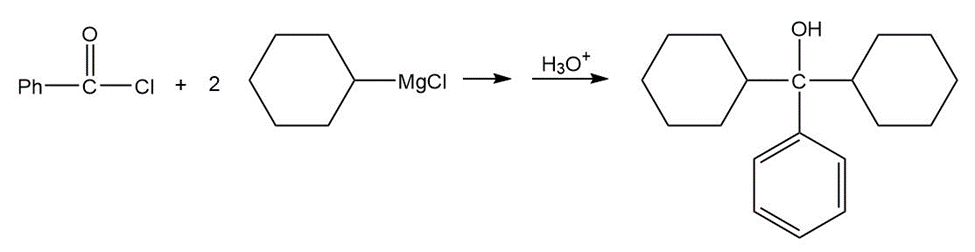
c.
Learning Materials
Features
Discover
Chapter 10: Q17P (page 519)
Show how you would add Grignard reagents to acid chlorides or esters to synthesize the following alcohols.
a.
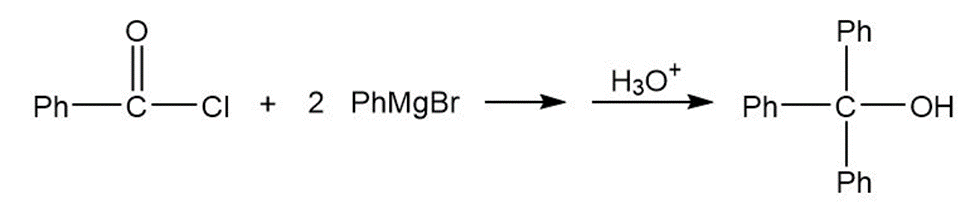
b.

c.

All the tools & learning materials you need for study success - in one app.
Get started for free
Show how you would synthesize the following compounds from any starting materials containing no more than six carbon atoms.
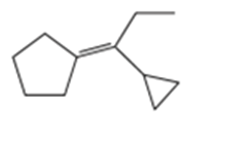
a)
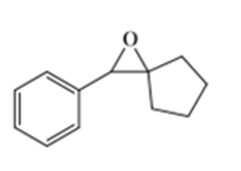
b)
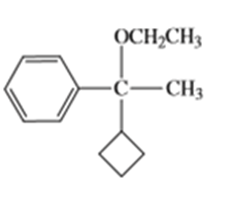
c)
A nitro group (-NO2) effectively stabilizes a negative charge on an adjacent carbon atom through resonance:
Two of the following nitrophenols are much more acidic than phenol itself. The third compound is only slightly more acidic than phenol. Use resonance structures of the appropriate phenoxide ions to show why two of these anions should be unusually stable.
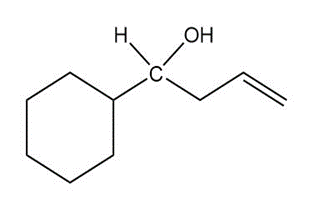
Question: Show two ways you could synthesize each of the following secondary alcohols by adding an appropriate Grignard reagent to an aldehyde.
a.
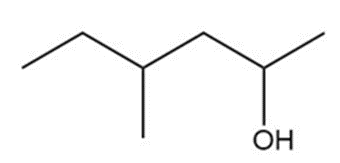
b.
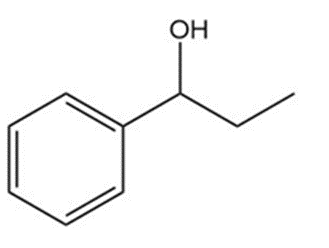
c.
Give a systematic (IUPAC) name for each diol.
a.
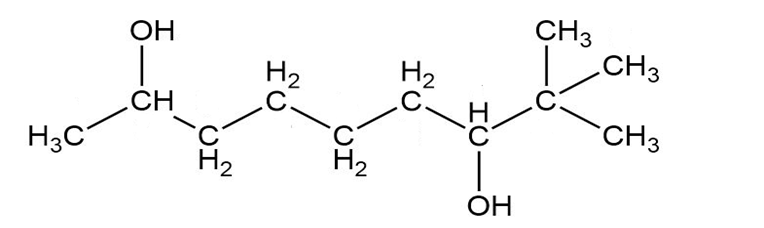
b.
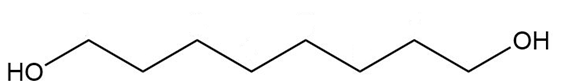
c.
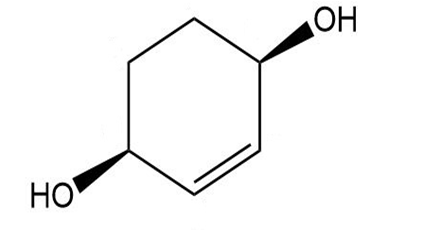
d.
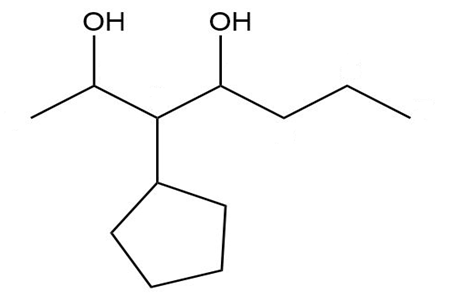
e.
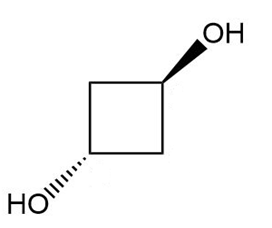
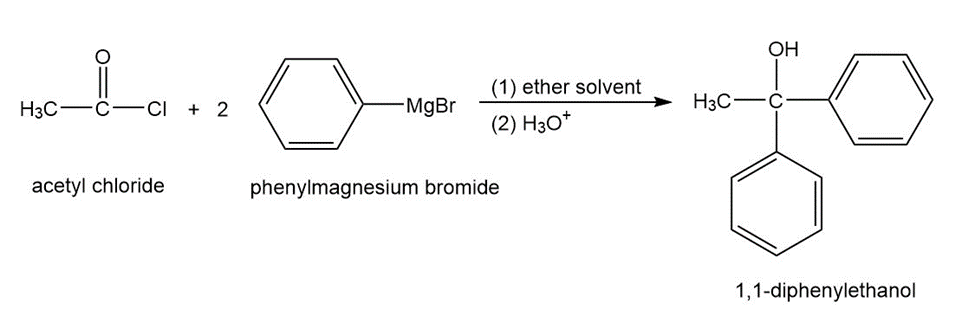
Propose a mechanism for the reaction of acetyl chloride with phenylmagnesium bromide to give 1,1-diphenylethanol.
What do you think about this solution?
We value your feedback to improve our textbook solutions.