Chapter 10: Q44P (page 538)
Show how you would synthesize the following compounds from any starting materials containing no more than six carbon atoms.

a)
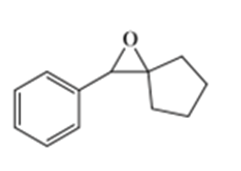
b)

c)
Short Answer

a)
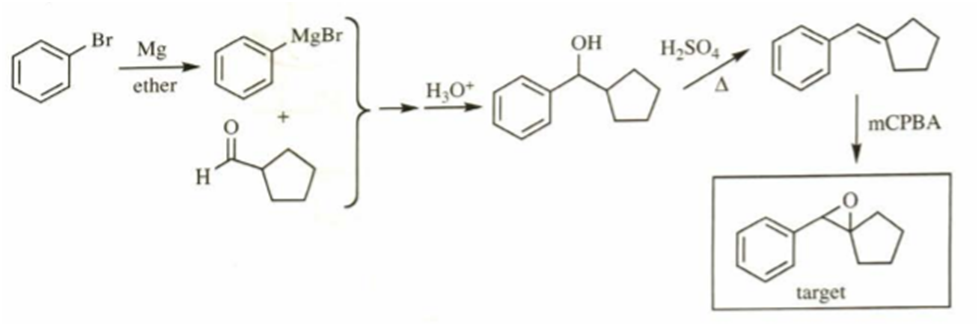
b)
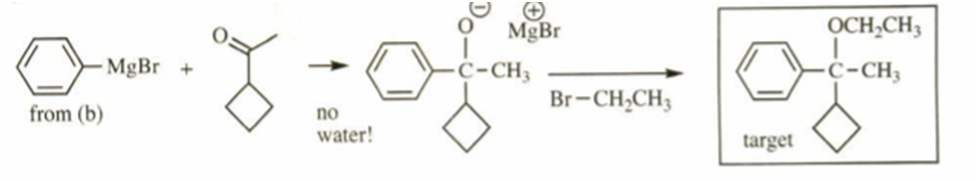
c)
Learning Materials
Features
Discover
Chapter 10: Q44P (page 538)
Show how you would synthesize the following compounds from any starting materials containing no more than six carbon atoms.

a)

b)

c)

a)

b)

c)
All the tools & learning materials you need for study success - in one app.
Get started for free
A formate ester, such as ethyl formate, reacts with an excess of a Grignard reagent to give (after protonation) secondary alcohols with two identical alkyl groups.
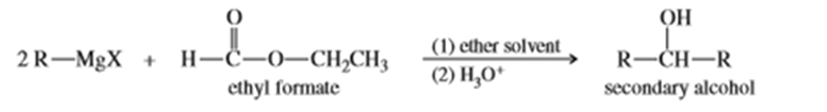
a. Propose a mechanism to show how the reaction of ethyl formate with an excess of allylmagnesium bromide gives, after protonation, hepta-1,6-dien-4-ol.
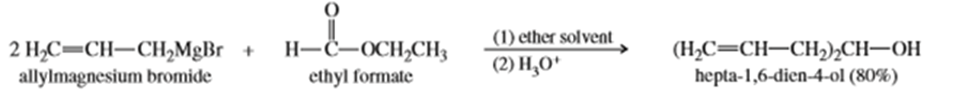
b. Show how you would use reactions of Grignard reagents with ethyl formate to synthesize the following secondary alcohols.
i. pentan-3-ol
ii. diphenylmethanol
iii. trans, trans-nona-2,7-dien-5-ol
Give a systematic (IUPAC) name for each diol.
a.
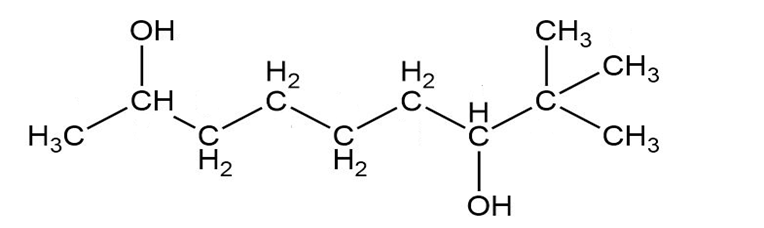
b.
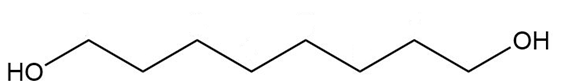
c.
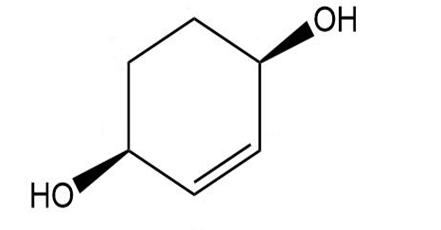
d.
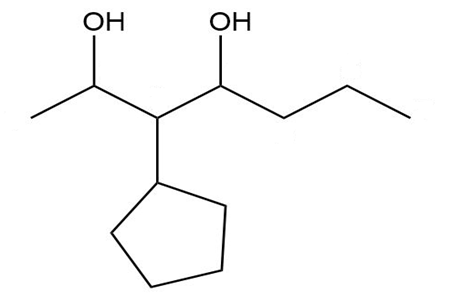
e.

Suggest carbonyl compounds and reducing agents that might be used to form the following alcohols.

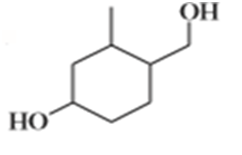
e)
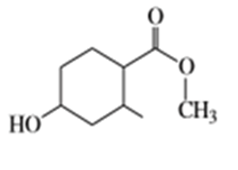
f)
Many hunting dogs enjoy standing nose-to-nose with a skunk while barking furiously, oblivious to the skunk spray directed toward them. One moderately effective way of lessening the amount of odor is to wash the dog in a bath containing dilute hydrogen peroxide, sodium bicarbonate, and some mild dish detergent. Use chemical reactions to describe how this mixture helps to remove the skunk spray from the dog. The two major components of skunk oil are 3-methylbutane-1-thiol and but-2-ene-1-thiol.
For each synthesis, start with bromocyclohexane and predict the products. Assume that an excess of each reactant is added so that all possible reactions that can happen will happen.
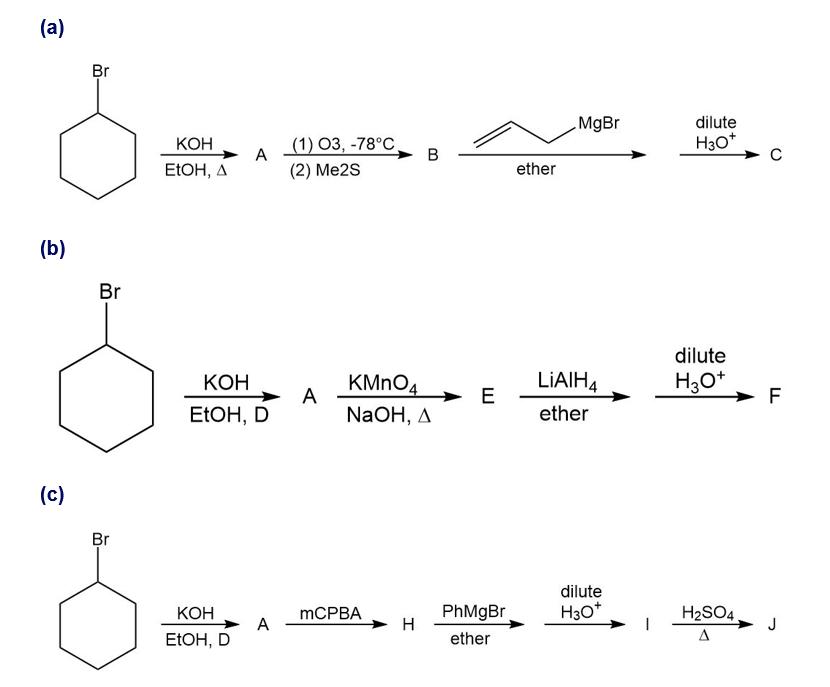
What do you think about this solution?
We value your feedback to improve our textbook solutions.