Chapter 10: Q15P (page 517)
Show how you would synthesize each tertiary alcohol by adding an appropriate Grignard reagent to a ketone.
(a)3-phenylhexane-3-ol (3ways)
(b) Ph3 COH
(c) 1-ethylcyclopentanol
(d) 2-cyclopentylpentan-2-ol
Learning Materials
Features
Discover
Chapter 10: Q15P (page 517)
Show how you would synthesize each tertiary alcohol by adding an appropriate Grignard reagent to a ketone.
(a)3-phenylhexane-3-ol (3ways)
(b) Ph3 COH
(c) 1-ethylcyclopentanol
(d) 2-cyclopentylpentan-2-ol
All the tools & learning materials you need for study success - in one app.
Get started for free
Suggest carbonyl compounds and reducing agents that might be used to form the following alcohols.
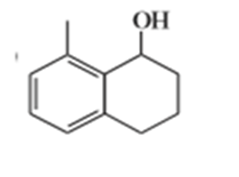

e)
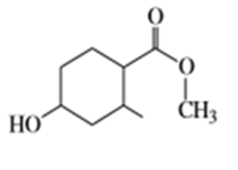
f)
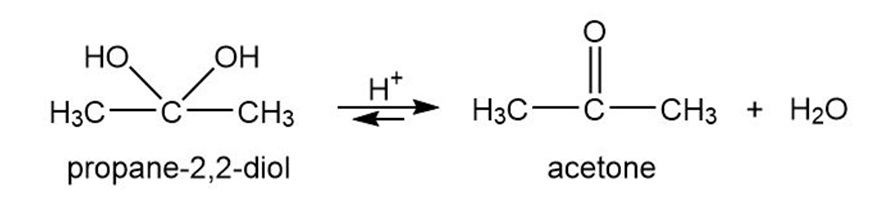
Geminal diols, or 1,1-diols, are usually unstable, spontaneously losing water to give carbonyl compounds. Therefore, germinal diols are regarded as hydrated forms of ketones and aldehydes. Propose a mechanism for the acid-catalyzed loss of water from propane-2,2-diol to give acetone.
Predict the products of the following reactions.
(a) sec-butylmagnesium iodide + D2O
(b) n-butyllithium + CH3CH2OH
(c) isobutylmagnesium bromide + but-1-yne
(d)

(e)

Predict which member of each pair will be more acidic. Explain your answers.
(a) methanol or tert-butyl alcohol
(b)2-chloropropan-1-ol or 3-chloropropan-1-ol
(c)2-chloroethanol or 2,2-dichloroethanol
(d)2,2-dichloropropan-1-ol or 2,2-difluoropropan-1-ol
A formate ester, such as ethyl formate, reacts with an excess of a Grignard reagent to give (after protonation) secondary alcohols with two identical alkyl groups.
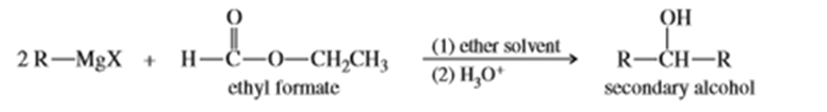
a. Propose a mechanism to show how the reaction of ethyl formate with an excess of allylmagnesium bromide gives, after protonation, hepta-1,6-dien-4-ol.
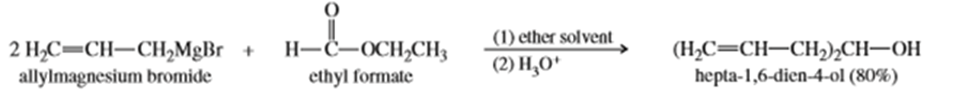
b. Show how you would use reactions of Grignard reagents with ethyl formate to synthesize the following secondary alcohols.
i. pentan-3-ol
ii. diphenylmethanol
iii. trans, trans-nona-2,7-dien-5-ol
What do you think about this solution?
We value your feedback to improve our textbook solutions.