Chapter 14: Q 39 P (page 747)
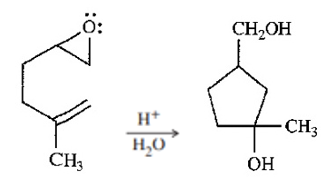
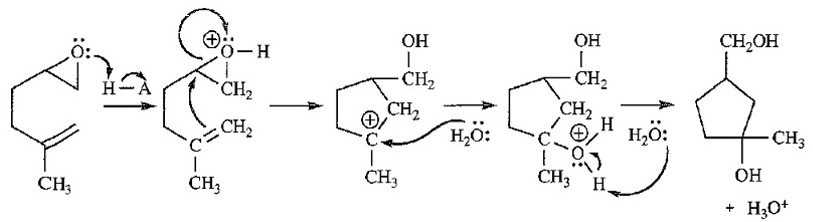
Question. The following reaction resembles the acid-catalyzed cyclization of squalene oxide. Propose a mechanism for this reaction.
Short Answer
Learning Materials
Features
Discover
Chapter 14: Q 39 P (page 747)
Question. The following reaction resembles the acid-catalyzed cyclization of squalene oxide. Propose a mechanism for this reaction.


All the tools & learning materials you need for study success - in one app.
Get started for free
Show how the following ethers might be synthesized using (1) alkoxymercuration-demercuration and (2) the Williamson synthesis. (When one of these methods cannot be used for the given ether, point out why it will not work.)
Show how you would use a protecting group to convert 4-bromobutan-1-ol to hept-5-yn-1-ol.
Show how you would accomplish the following transformations. Some of these examples require more than one step.

Question. Propose a complete mechanism for the reaction of cyclopentene oxide with sodium methoxide in methanol.
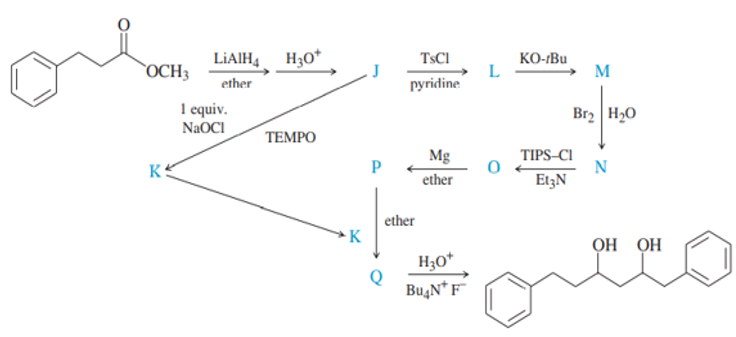
Question. Give the structures of the intermediates represented by letters in this synthesis.
What do you think about this solution?
We value your feedback to improve our textbook solutions.