Chapter 14: Q 43P (page 748)
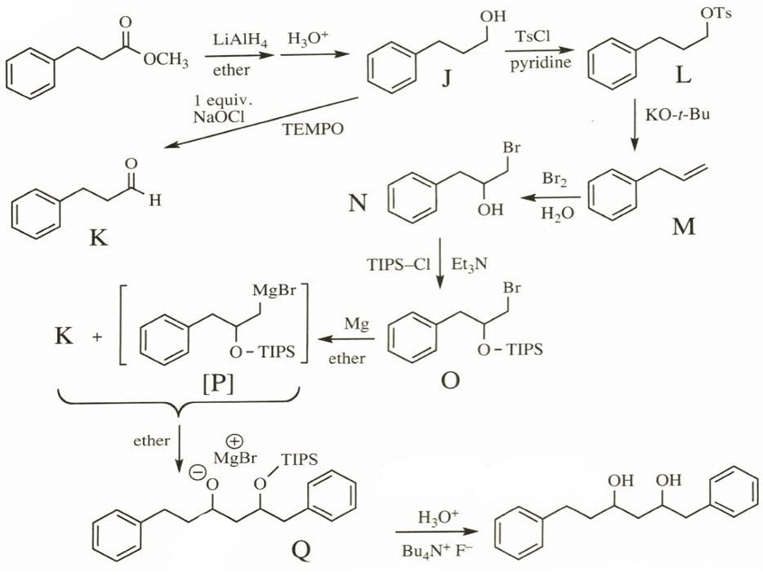
Question. Give the structures of the intermediates represented by letters in this synthesis.
Short Answer
Learning Materials
Features
Discover
Chapter 14: Q 43P (page 748)
Question. Give the structures of the intermediates represented by letters in this synthesis.


All the tools & learning materials you need for study success - in one app.
Get started for free
In 2012, a group led by Professor Masayuki Satake of the University of Tokyo reported the isolation and structure determination of a toxin from a marine algal bloom that decimated the fish population off the New Zealand coast in 1998. Extensive mass spectrometry and NMR experiments ultimately led to the structure shown below, named Brevisulcenal-F. (See Journal of the American Chemical Society, 2012, 134, 4963-4968.) This structure holds the record for the largest number of fused rings, at 17.
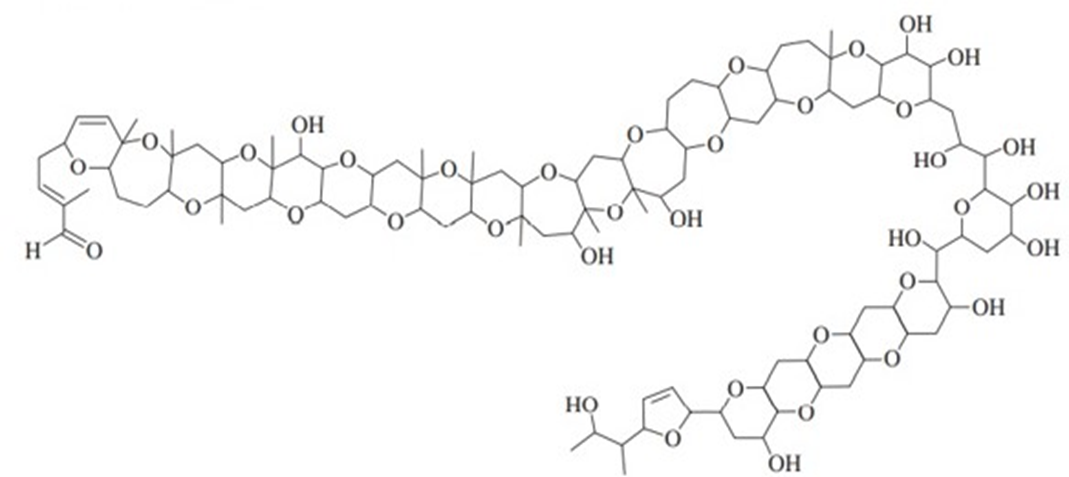
Brevisulcenal-F
a. How many ether groups are present?
b. How many alcohol groups are present? Classify the alcohols as 1o or 2o or 3o.
c. Are there any other oxygen-containing functional groups? Which, if any?
Which of the following ethers can be formed in good yield by condensation of the corresponding alcohols? For those that cannot be formed by condensation, suggest an alternative method that will work.
(a)
(b)
(c)
Question. (a) Show how you would synthesize the pure (R) enantiomer of 2-pentyl methyl sulfide, starting with pure (R)-pentan-2-ol and any reagents you need.
(b) Show how you would synthesize the pure (S) enantiomer of the product, still starting with (R)-pentan-2-ol and any reagents you need.
1,4-Dioxane is made commercially by the acid-catalyzed condensation of an alcohol.
(a)Show what alcohol will undergo condensation, with loss of water, to give 1,4-dioxane.
(b)Propose a mechanism for this reaction
Question: (a) Predict the values of m/zand the structures of the most abundant fragments you would observe in the mass spectrum of di-n-propyl-ether.
(b) Give logical fragmentations to account for the following ions observed in the mass spectrum of 2-methoxypentane: 102,87,71,59,31.
What do you think about this solution?
We value your feedback to improve our textbook solutions.