Chapter 14: Q13P (page 721)
Which of the following ethers can be formed in good yield by condensation of the corresponding alcohols? For those that cannot be formed by condensation, suggest an alternative method that will work.
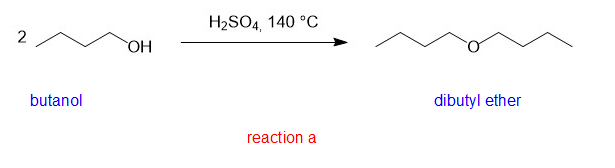
(a)
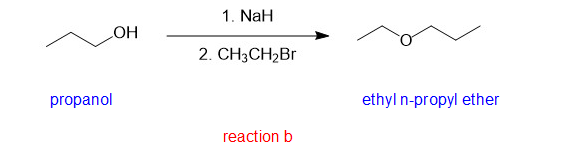
(b)
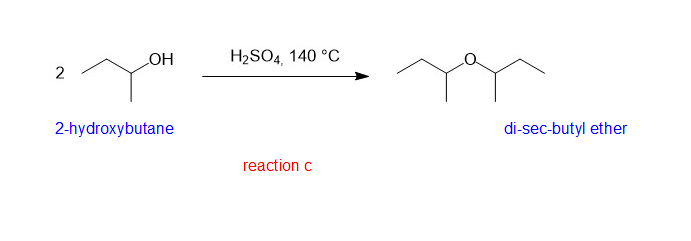
(c)
Short Answer
(a)is a symmetrical ether and hence good yield can be produced by condensation method.
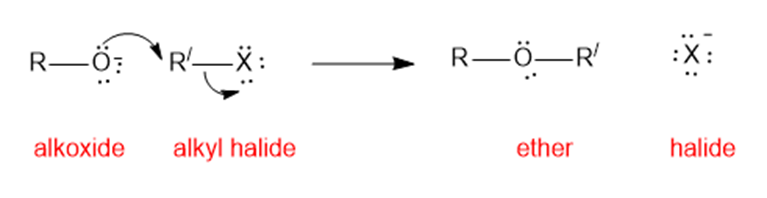
(b)is an unsymmetrical ether and hence good yield cannot be produced by condensation method. In such case, Williamson ether synthesis can eb used for good yields.
(c) is a symmetrical ether and hence good yield can be produced by condensation method.