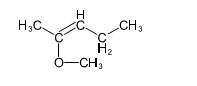
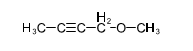Chapter 14: Q 37P (page 747)
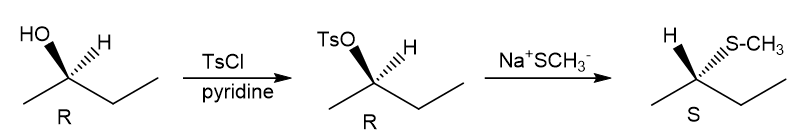
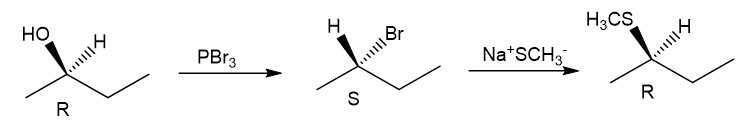
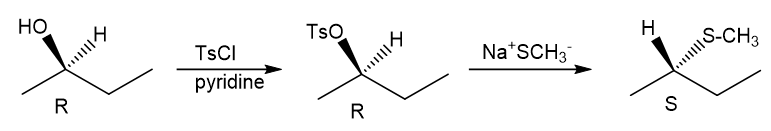
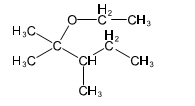
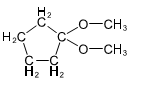
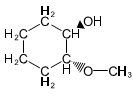
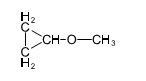
Question. (a) Show how you would synthesize the pure (R) enantiomer of 2-pentyl methyl sulfide, starting with pure (R)-pentan-2-ol and any reagents you need.
(b) Show how you would synthesize the pure (S) enantiomer of the product, still starting with (R)-pentan-2-ol and any reagents you need.
Short Answer
(a)
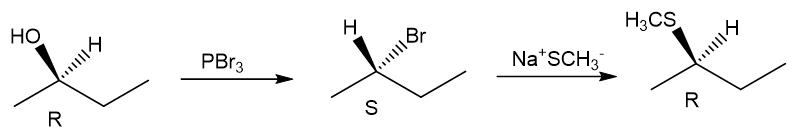
(b)