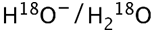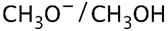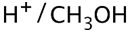Chapter 14: Q 36 P (page 747)
Question. Grignard reactions are often limited by steric hindrance. While Grignard reagents react in high yield with ethylene oxide and monosubstituted epoxides, yields are often lower with disubstituted epoxides. Tri- and tetrasubstituted epoxides react with difficulty, if at all.
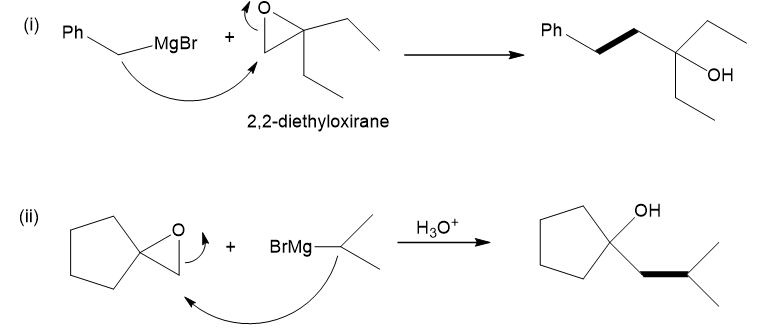
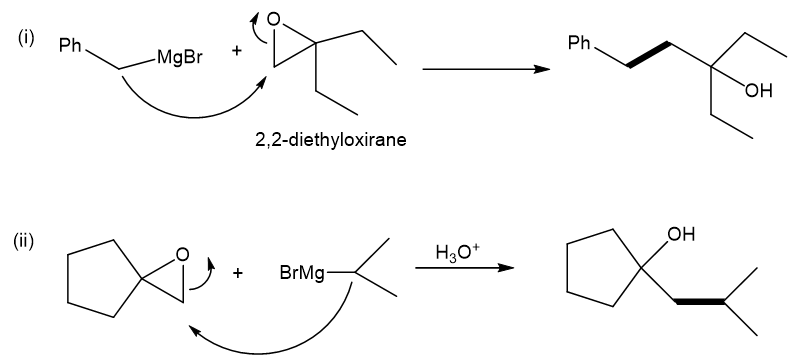
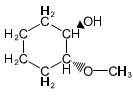
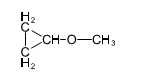
(a) Show how to make these alcohols by a Grignard reacting with an epoxide.
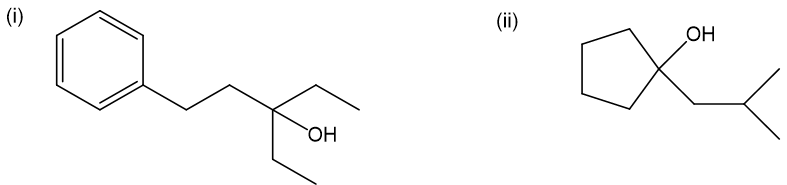
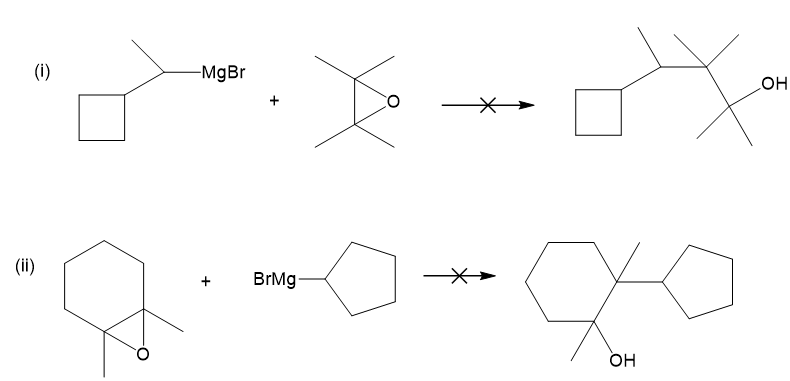
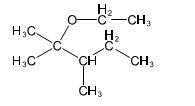
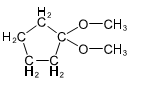
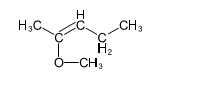
(b) These alcohols cannot be made by a Grignard plus an epoxide. Show the reagents that would be required and why that reaction would be unlikely to succeed.

Short Answer
(a)
(b)
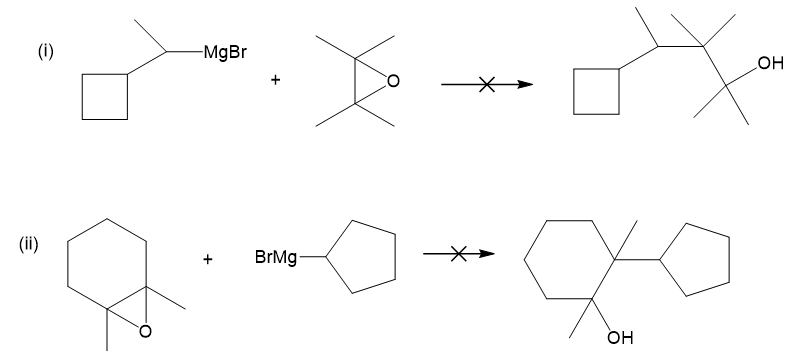
In each case, the epoxide is derived from a tetrasubstituted double bond, far too hindered for the Grignard reagent to approach.



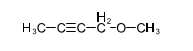
 (oxygen-labelled water)
(oxygen-labelled water)