Chapter 14: Q19P (page 729)
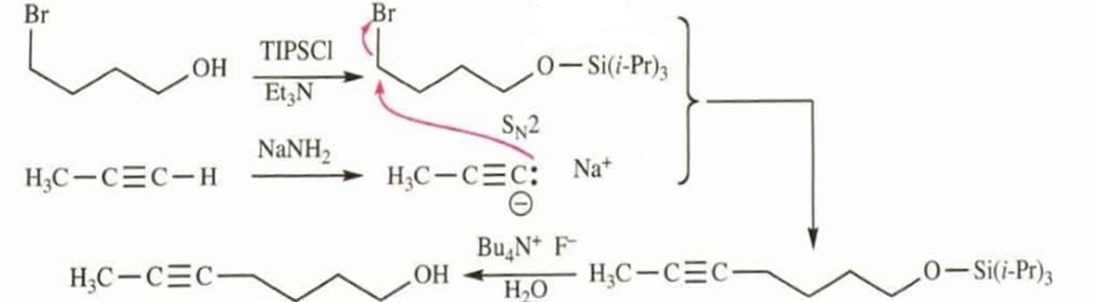
Show how you would use a protecting group to convert 4-bromobutan-1-ol to hept-5-yn-1-ol.
Short Answer
Learning Materials
Features
Discover
Chapter 14: Q19P (page 729)
Show how you would use a protecting group to convert 4-bromobutan-1-ol to hept-5-yn-1-ol.

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Show how you would convert pent-1-eneto each of the following compounds. You may use any additional reagents and solvents you need.
(a) 2-methoxypentane
(b) 1-methoxypentane
(c) 1-methoxypentan-2-ol
(d) 2-methoxypentan-1-ol
(e) 1-phenylpentan-2-ol
(f) 2-methoxy-1-phenylpentane
Show how the following ethers might be synthesized using (1) alkoxymercuration-demercuration and (2) the Williamson synthesis. (When one of these methods cannot be used for the given ether, point out why it will not work.)
Boron tribromidecleaves ethers to give alkyl halides and alcohols.
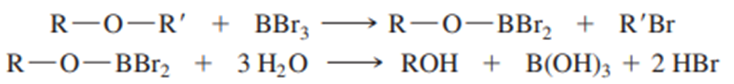
The reaction is thought to involve attack by a bromide ion on the Lewis acid-base adduct of the ether with(a strong Lewis acid). Propose a mechanism for the reaction of butyl methyl ether withto give (after hydrolysis) butan-1-ol and bromomethane.
(a) Tetramethyloxirane is too hindered to undergo nucleophilic substitution by the hindered alkoxide, potassium tert-butoxide. Instead, the product is the allylic alcohol shown. Propose a mechanism to explain this reaction. What type of mechanism does it follow?
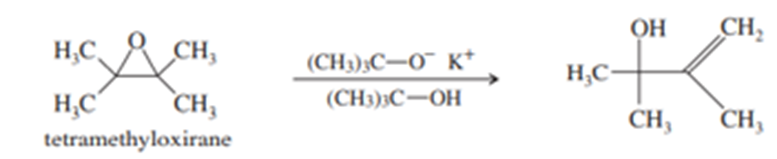
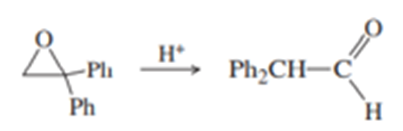
(b) Under mild acid catalysis, 1,1-diphenyloxirane undergoes a smooth conversion to diphenylethanal (diphenylacetaldehyde). Propose a mechanism for this reaction. (Hint: Think Pinacol.)
Propose a Williamson synthesis of 3-butoxy-1,1-dimethylcyclohexane from 3,3-dimethyl-cyclohexanol and butan-1-ol.
What do you think about this solution?
We value your feedback to improve our textbook solutions.