Chapter 14: Q1P. (page 711)
Rank the given solvents in decreasing order of their ability to dissolve each compound
(a) HCOO-Na+
(b)
(c)
Solvents
Ethyl ether, water, ethanol, dichloromethane
Short Answer
(1) Decreasing order of solvents’ ability to dissolve sodium acetate:
Water >ethanol > ethyl ether>dichloromethane
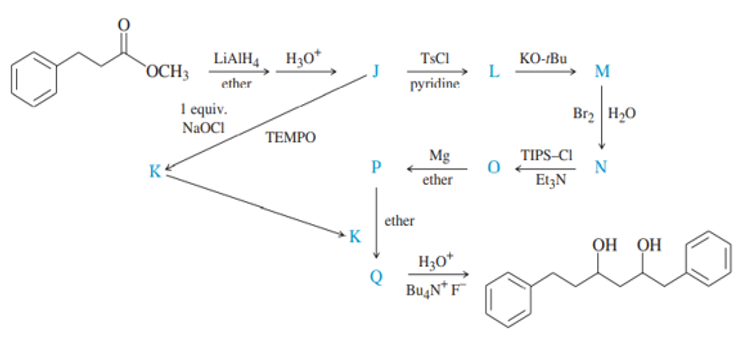
(2) Decreasing order of solvents’ ability to dissolve 2-methylnapthalene:
Dichloromethane >ethyl ether >ethanol >water
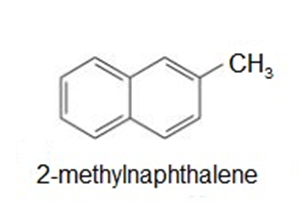
(3) Decreasing order of solvents’ ability to dissolve 1-napthol:Dichloromethane>ethyl ether>ethanol>water