Chapter 14: Q18P (page 728)
Mustard gas, , was used as a poisonous chemical agent in World War I. Mustard gas is much more toxic than a typical primary alkyl chloride. Its toxicity stems from its ability to alkylate amino groups on important metabolic enzymes, rendering the enzymes inactive.
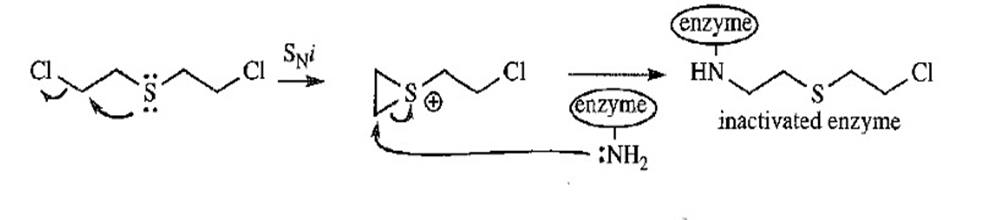
- Propose a mechanism to explain why mustard gas is an exceptionally potent alkylating agent.
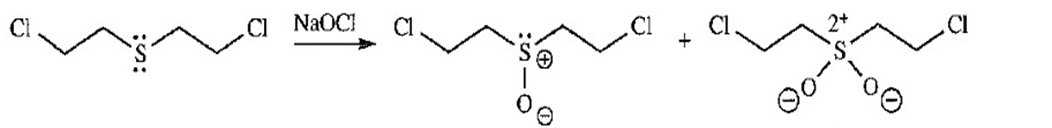
- Bleach (sodium hypochlorite,, a strong oxidizing agent) neutralizes and inactivates mustard gas. Bleach is also effective on organic stains because it oxidizes colored compounds to colorless compounds. Propose products that might be formed by the reaction of mustard gas with bleach.
Short Answer
a.
b.
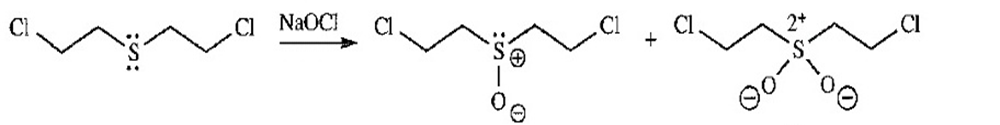
Mustard gas sulfoxide sulfone
 Mustard gas as potent alkylating agent
Mustard gas as potent alkylating agent