Chapter 14: Q25P (page 737)
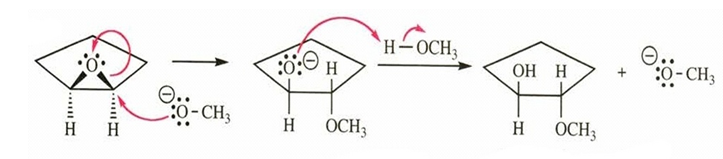
Question. Propose a complete mechanism for the reaction of cyclopentene oxide with sodium methoxide in methanol.
Short Answer
Answer.
Learning Materials
Features
Discover
Chapter 14: Q25P (page 737)
Question. Propose a complete mechanism for the reaction of cyclopentene oxide with sodium methoxide in methanol.
Answer.

All the tools & learning materials you need for study success - in one app.
Get started for free
Show the rest of the mechanism for formation of cyclized intermediate in figure 14-6.
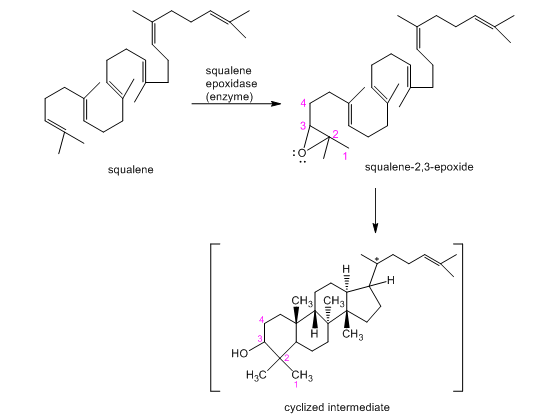
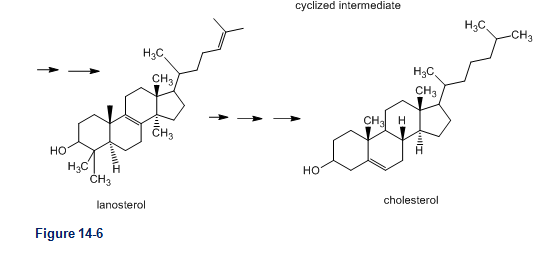
Role of squalene in the biosynthesis of steroids. The biosynthesis of steroids starts with epoxidation of squalene to squalene-2,3-epoxide. The opening of this epoxide promotes cyclization of the carbon skeleton under the control of an enzyme. The cyclized intermediate is converted to lanosterol and then to other steroids.
Question. (A true story.) An inexperienced graduate student moved into a laboratory and began work. He needed some diethyl ether for a reaction, so he opened an old, rusty 1-gallon can marked “ethyl ether” and found there was half a gallon left. To purify the ether, the student set up a distillation apparatus, started a careful distillation, and went to the stockroom for the other reagents he needed. While he was at the stockroom, the student heard a muffled “boom”. He quickly returned to his lab to find a worker from another laboratory putting out a fire. Most of the distillation apparatus was embedded in the ceiling.
(a) Explain what probably happened.
(b) Explain how this near disaster might have been prevented.
Propose a Williamson synthesis of 3-butoxy-1,1-dimethylcyclohexane from 3,3-dimethyl-cyclohexanol and butan-1-ol.
(a) Tetramethyloxirane is too hindered to undergo nucleophilic substitution by the hindered alkoxide, potassium tert-butoxide. Instead, the product is the allylic alcohol shown. Propose a mechanism to explain this reaction. What type of mechanism does it follow?
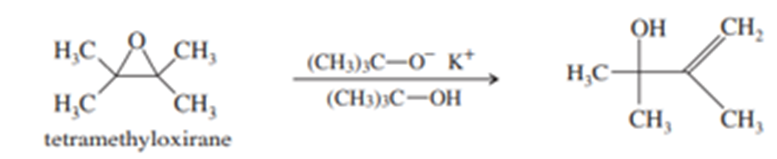
(b) Under mild acid catalysis, 1,1-diphenyloxirane undergoes a smooth conversion to diphenylethanal (diphenylacetaldehyde). Propose a mechanism for this reaction. (Hint: Think Pinacol.)
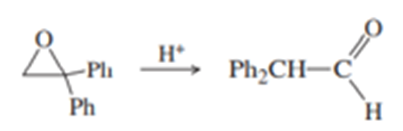
Question. Propose a mechanism for the following reaction.
What do you think about this solution?
We value your feedback to improve our textbook solutions.