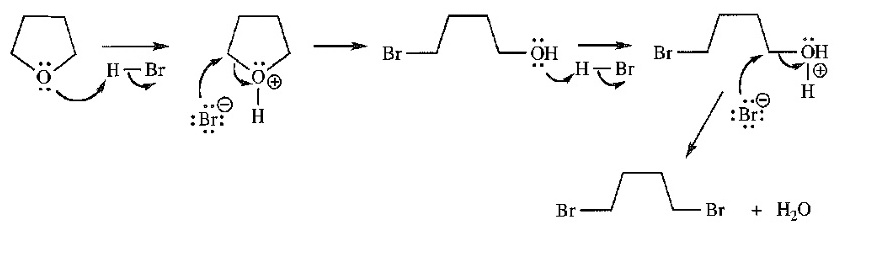
Chapter 14: Q14P. (page 723)
Question. Propose a mechanism for the following reaction.
Short Answer
Answer
Learning Materials
Features
Discover
Chapter 14: Q14P. (page 723)
Question. Propose a mechanism for the following reaction.
Answer

All the tools & learning materials you need for study success - in one app.
Get started for free
Propylene oxide is a chiral molecule. Hydrolysis of propylene oxide gives propylene glycol, another chiral molecule.
(a) Draw the enantiomers of propylene oxide.
(b) Propose a mechanism for the acid-catalyzed hydrolysis of pure .
(c) Propose a mechanism for the base-catalyzed hydrolysis of pure .
(d) Explain why the acid-catalyzed hydrolysis of optically active propylene oxide gives a product with lower enantiomeric excess and a rotation opposite that of the product of the base- catalyzed hydrolysis.
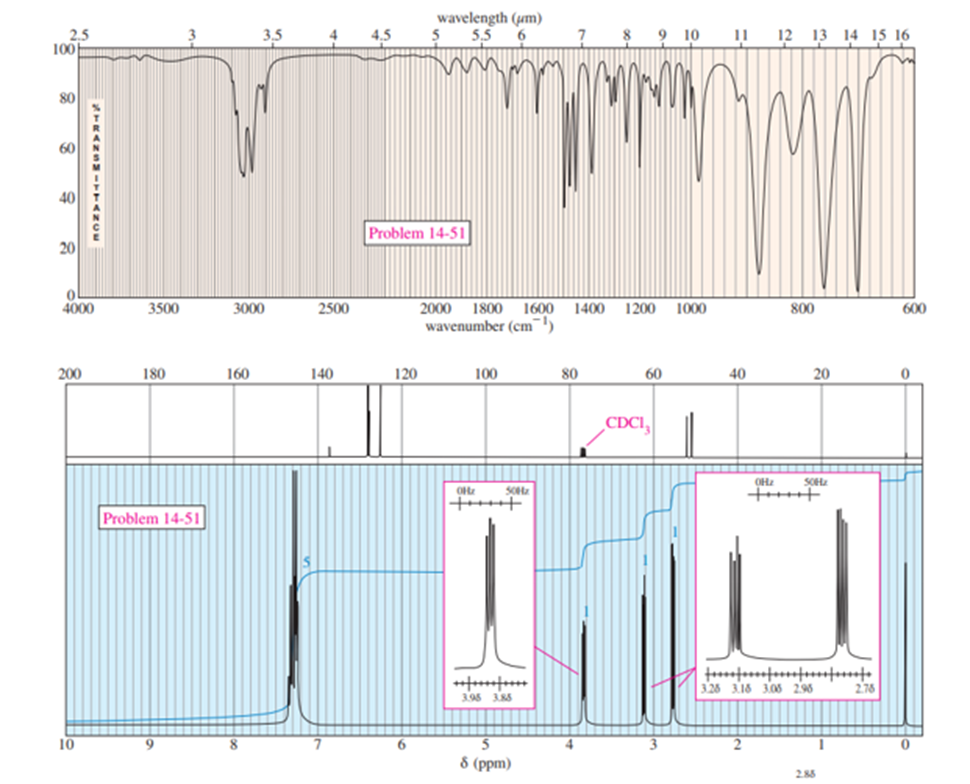
A compound of molecular formula C8H8Ogives the IR and NMR spectra shown here. Propose a structure and show how it is consistent with the observed absorptions.
Predict the major product when each reagent reacts with ethylene oxide.
(a) NaOCH2CH3(Sodium ethoxide)
(b) NaNH2(sodium amide)
(c) NaSPh (sodium thiophenoxide)
(d) PhNH2(aniline)
(e) KCN (potassium cyanide)
(f) NaN3(soidum azide)
1,4-Dioxane is made commercially by the acid-catalyzed condensation of an alcohol.
(a)Show what alcohol will undergo condensation, with loss of water, to give 1,4-dioxane.
(b)Propose a mechanism for this reaction
Which of the following ethers can be formed in good yield by condensation of the corresponding alcohols? For those that cannot be formed by condensation, suggest an alternative method that will work.
(a)
(b)
(c)
What do you think about this solution?
We value your feedback to improve our textbook solutions.