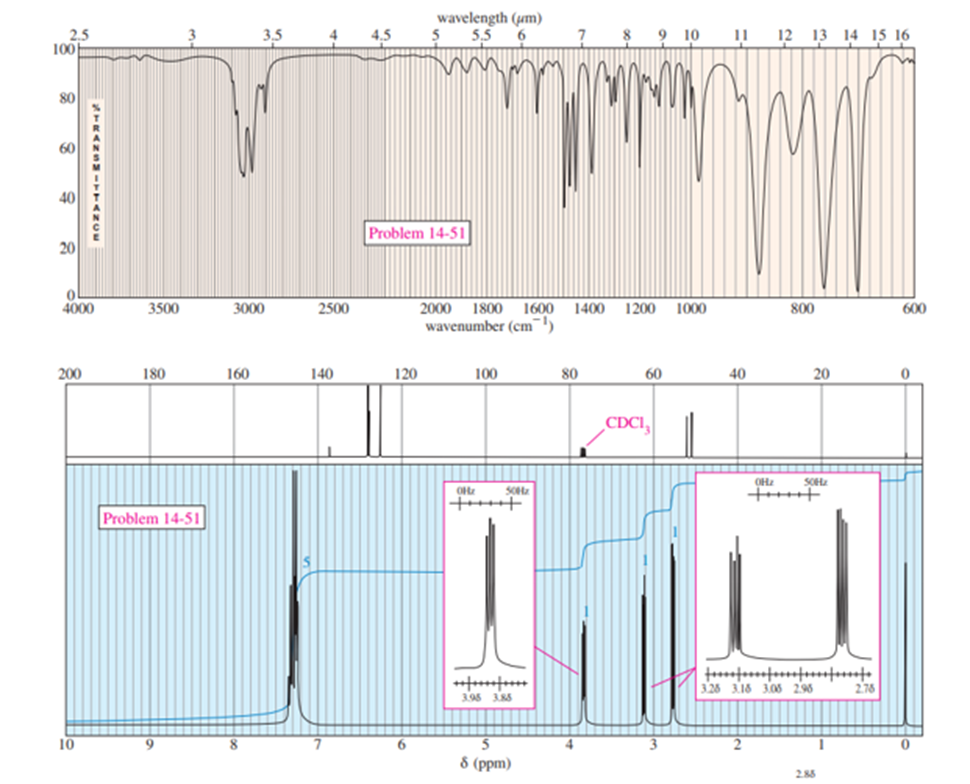
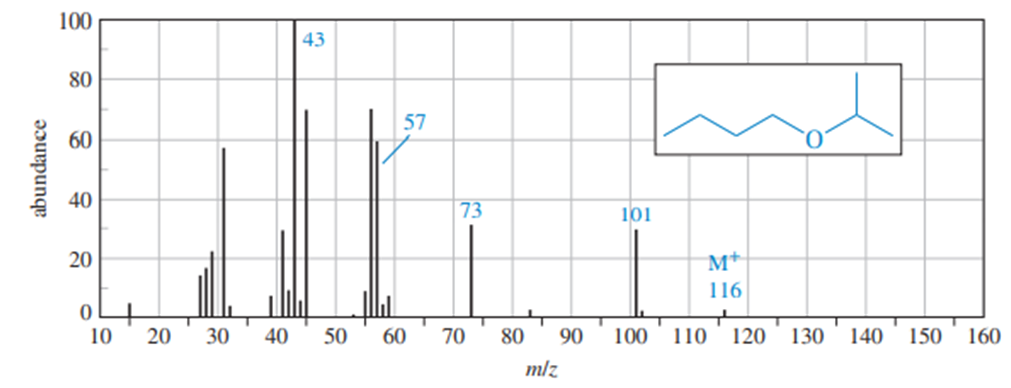
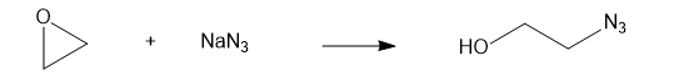Chapter 14: 14-26P (page 737)
Predict the major product when each reagent reacts with ethylene oxide.
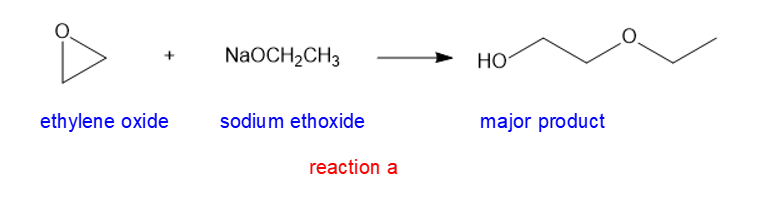
(a) NaOCH2CH3(Sodium ethoxide)
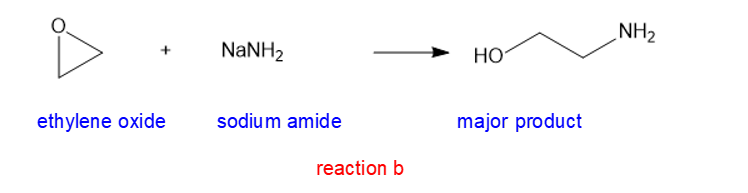
(b) NaNH2(sodium amide)
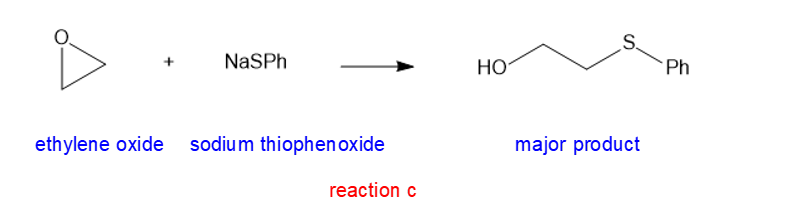
(c) NaSPh (sodium thiophenoxide)
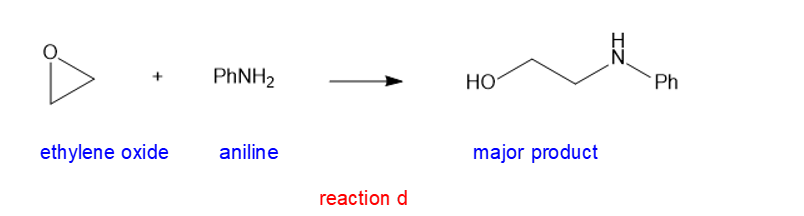
(d) PhNH2(aniline)
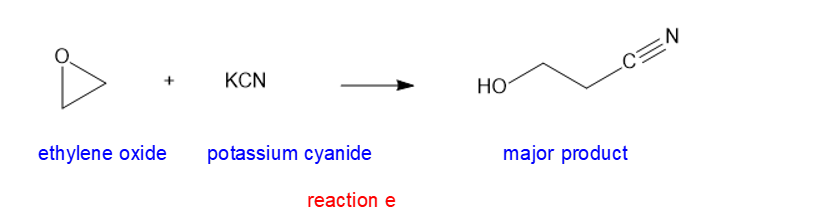
(e) KCN (potassium cyanide)
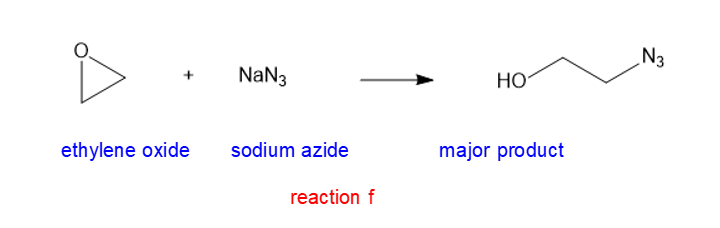
(f) NaN3(soidum azide)
Short Answer
(a)
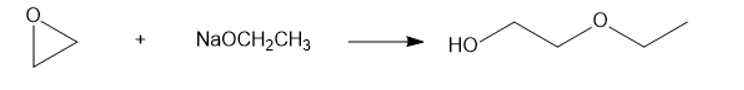
(b)

(c)
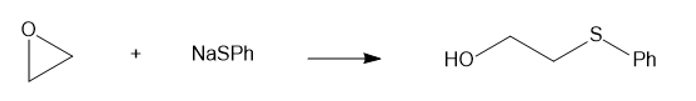
(d)
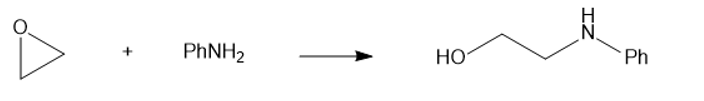
(e)
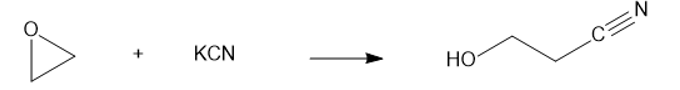
(f)