Chapter 14: Q7P (page 718)
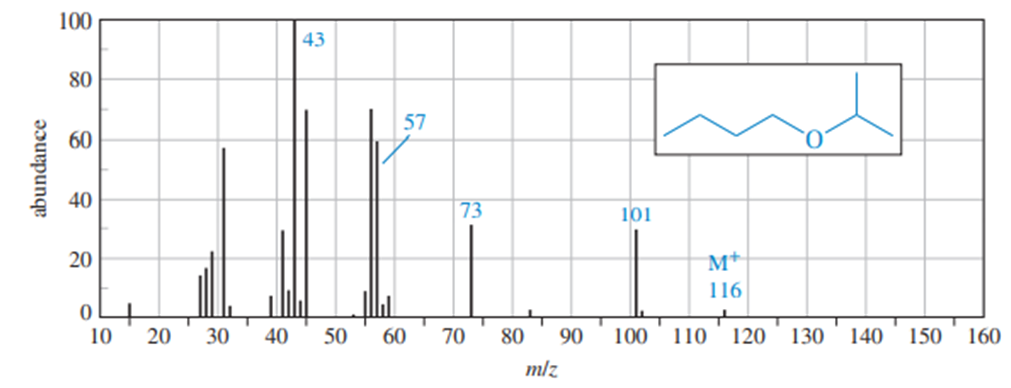
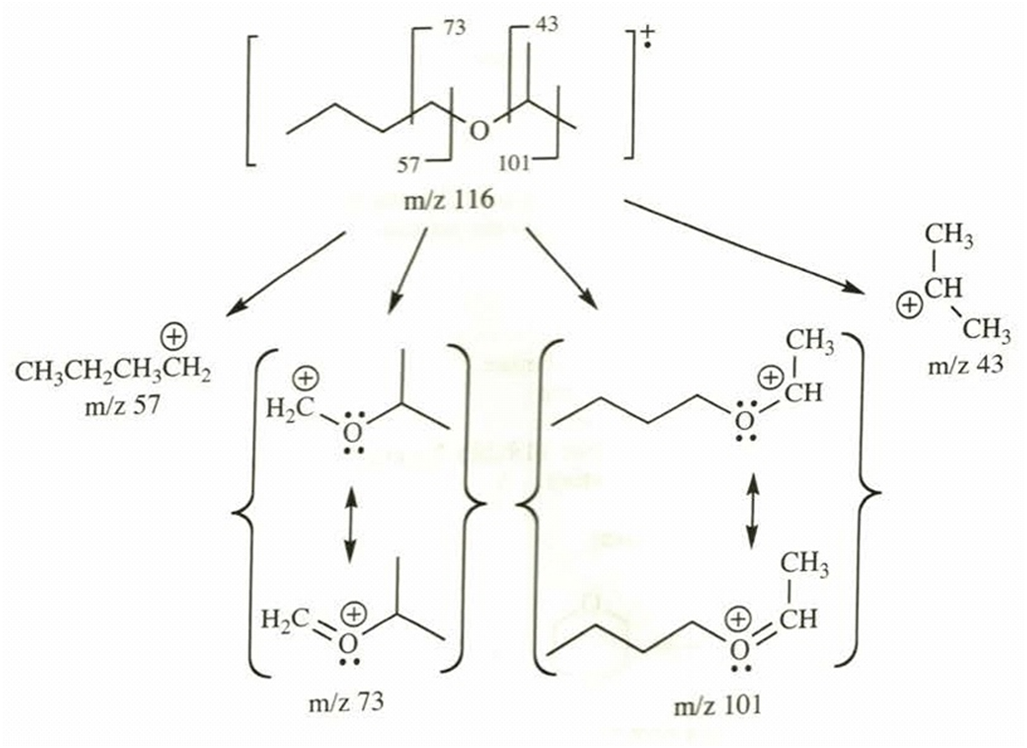
Propose a fragmentation to account for each numbered peak in the mass spectrum of n-butyl isopropyl ether.
Short Answer
Learning Materials
Features
Discover
Chapter 14: Q7P (page 718)
Propose a fragmentation to account for each numbered peak in the mass spectrum of n-butyl isopropyl ether.


All the tools & learning materials you need for study success - in one app.
Get started for free
Question. Grignard reactions are often limited by steric hindrance. While Grignard reagents react in high yield with ethylene oxide and monosubstituted epoxides, yields are often lower with disubstituted epoxides. Tri- and tetrasubstituted epoxides react with difficulty, if at all.
(a) Show how to make these alcohols by a Grignard reacting with an epoxide.
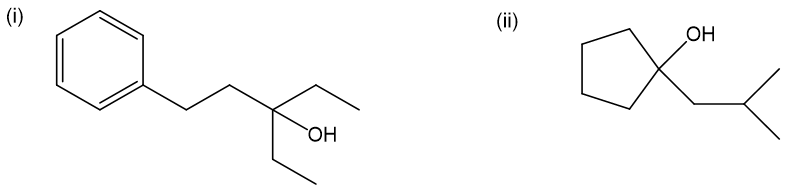
(b) These alcohols cannot be made by a Grignard plus an epoxide. Show the reagents that would be required and why that reaction would be unlikely to succeed.

Question. Predict the major products of the following reactions, including stereochemistry where appropriate.
 (oxygen-labelled water)
(oxygen-labelled water)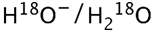
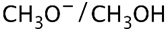
(2S,3R)-2-ethyl-2,3-dimethyloxirane +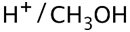
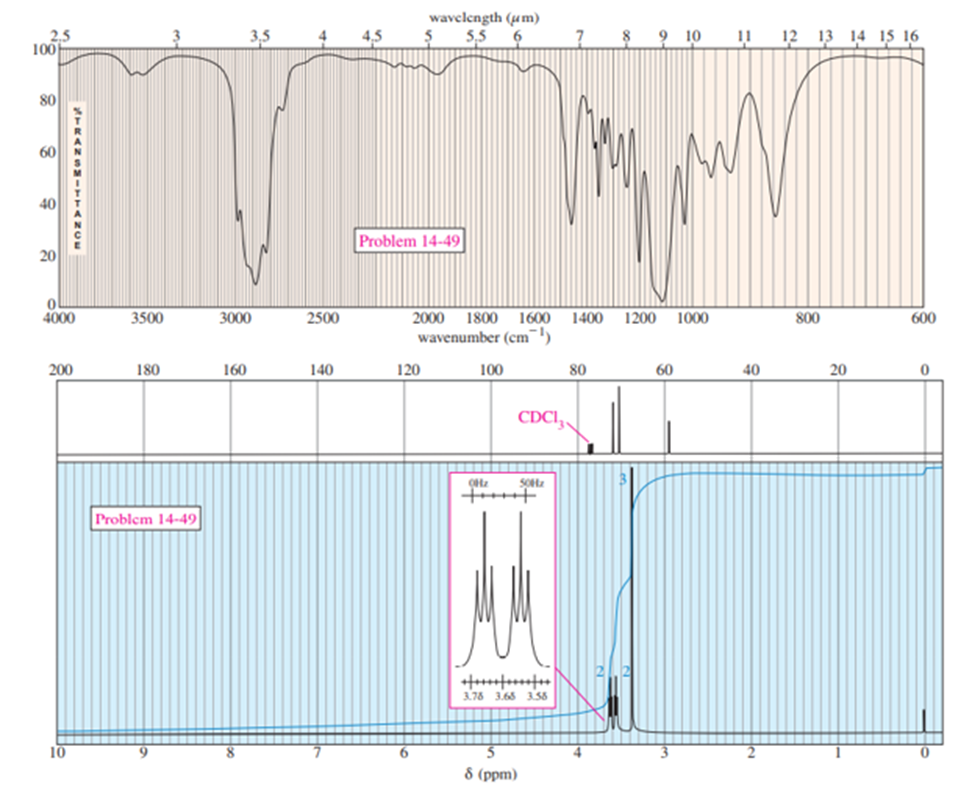
An acid-catalyzed reaction was carried out using methyl cellosolve (2-methoxyethanol) as the solvent. When the 2-methoxyethanol was redistilled, a higher-boiling point fraction (bp 162oC) was also recovered. The mass spectrum of this fraction showed the molecular weight to be 134. The IR and NMR spectrum are shown here. Determine the structure for this compound and propose a mechanism for its formation.
Question. Glycerol (propane-1,2,3-triol) is a viscous syrup with molecular weight 92g/mol, boiling point 290oC, and density 1.24g/mol. Transforming the three hydroxy groups into their trimethylsilyl ethers (using chlorotrimethylsilane and a tertiary amine) produces a liquid that flows easily, has molecular weight 309g/mol, boiling point approximately 180oC, and density 0.88g/mol. Draw the structures of these two compounds and explain why glycerol has a lower molecular weight but a much higher boiling point and density.
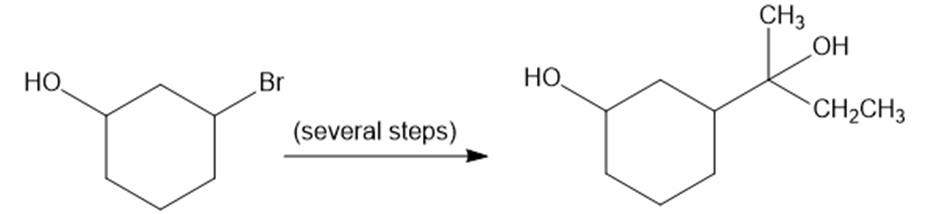
Show how you would convert 2-bromocyclohexanol to the following diol. You may use any additional reagents you need.
What do you think about this solution?
We value your feedback to improve our textbook solutions.