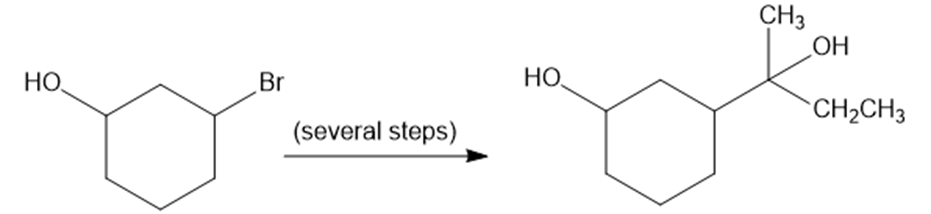
Chapter 14: Q 46P (page 748)
Show how you would convert 2-bromocyclohexanol to the following diol. You may use any additional reagents you need.
Short Answer
Learning Materials
Features
Discover
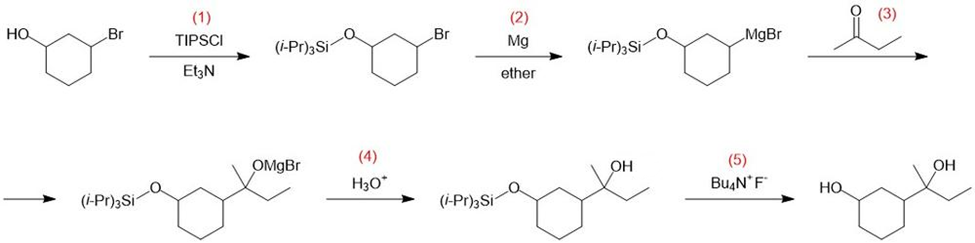
Chapter 14: Q 46P (page 748)
Show how you would convert 2-bromocyclohexanol to the following diol. You may use any additional reagents you need.


All the tools & learning materials you need for study success - in one app.
Get started for free
Question. Grignard reactions are often limited by steric hindrance. While Grignard reagents react in high yield with ethylene oxide and monosubstituted epoxides, yields are often lower with disubstituted epoxides. Tri- and tetrasubstituted epoxides react with difficulty, if at all.
(a) Show how to make these alcohols by a Grignard reacting with an epoxide.
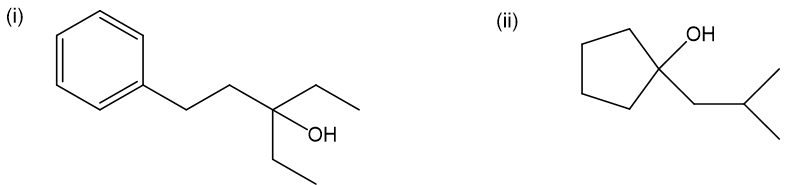
(b) These alcohols cannot be made by a Grignard plus an epoxide. Show the reagents that would be required and why that reaction would be unlikely to succeed.

Give the expected products of the following reactions. Include a protonation step where necessary.
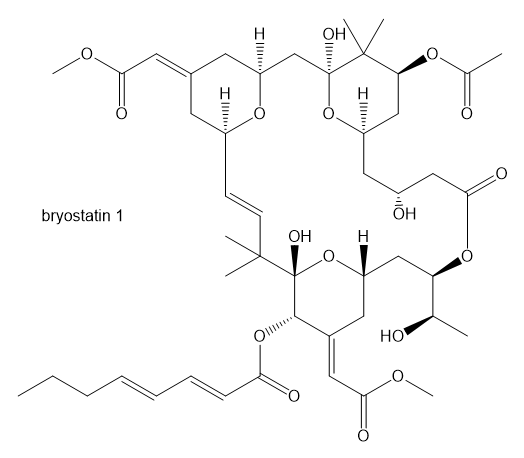
One of the crowning achievements of natural products synthesis was Bryostatin 1, published by Professor Gary Keck (University of Utah; Journal of the American Chemical Society,2011, 133, 744-747). The Bryostatins are a family of compounds isolated from aquatic invertebrates known as Bryozoans. The compounds are of interest for a variety of biological effects, including anti-cancer activity and reversing brain damage in rodents.
a. How many ether functional groups are present in Bryostatin 1?
b. Identify the other oxygen-containing functional groups.
c. This is called a macrolide because it contains a large number of atoms in the large ring. How many atoms are in the large ring?
d. How many chiral centers are in this molecule?
e. Using the number of chiral centers you reported in part (d), calculate the number of stereoisomers possible at these chiral centers. (Ignore stereoisomers at double bonds.)
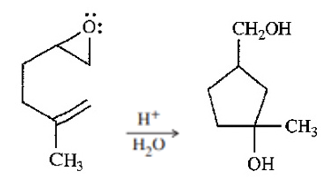
Question. The following reaction resembles the acid-catalyzed cyclization of squalene oxide. Propose a mechanism for this reaction.
Propose a Williamson synthesis of 3-butoxy-1,1-dimethylcyclohexane from 3,3-dimethyl-cyclohexanol and butan-1-ol.
What do you think about this solution?
We value your feedback to improve our textbook solutions.