Chapter 14: Q 47P (page 748)
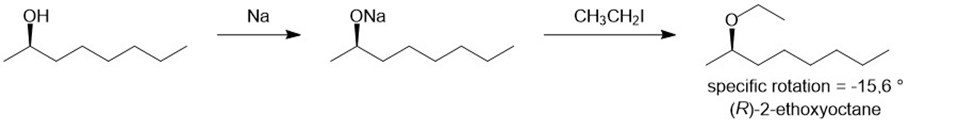
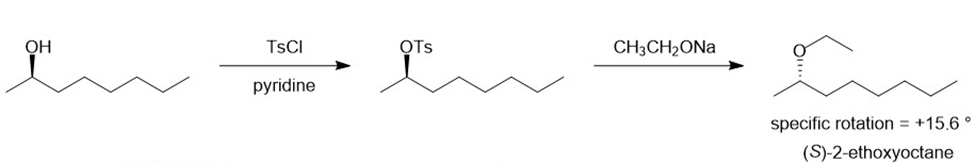
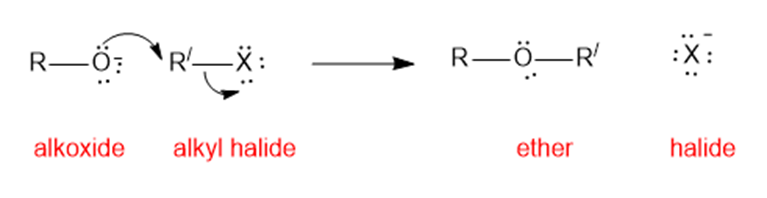
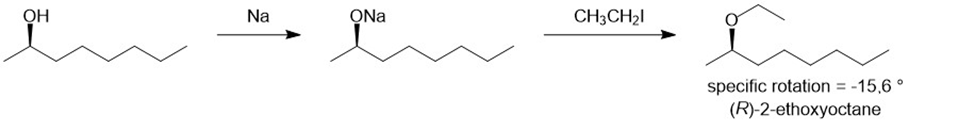
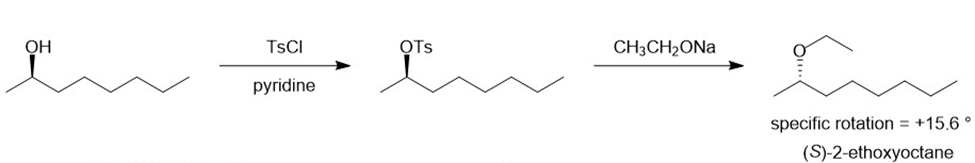
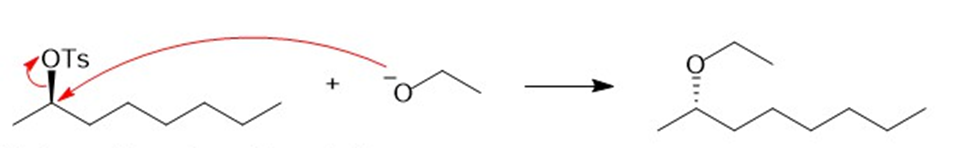
There are two ways of making 2-ethoxyoctane from octan-2-ol using the Williamson ether synthesis. When pure (-) -octan-2-ol of specific rotation -8.240is treated with sodium metal and then ethyl iodide, the product is 2-ethoxyoctane with a specific rotation of -15.60. When pure (-) -octan-2-ol is treated with tosyl chloride and pyridine and then with sodium ethoxide, the product is also 2-ethoxyoctane. Predict the rotation of the 2-ethoxyoctane made using the tosylation/sodium ethoxide procedure, and propose a detailed mechanism to support your prediction.
Short Answer