Chapter 14: Q 48P (page 749)
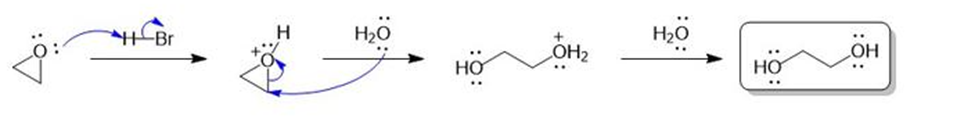
(a) When ethylene oxide is treated with anhydrous HBr gas, the major product is 1,2-dibromoethane. When ethylene oxide is treated with concentrated aqueous HBr, the major product is ethylene glycol. Use mechanisms to explain these results.
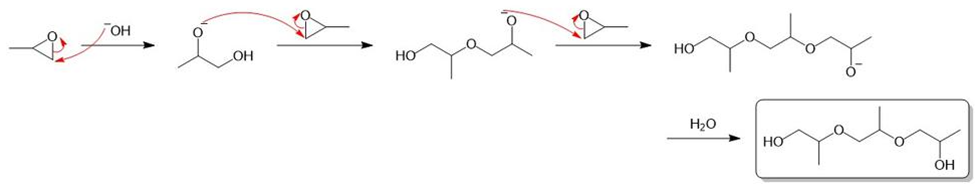
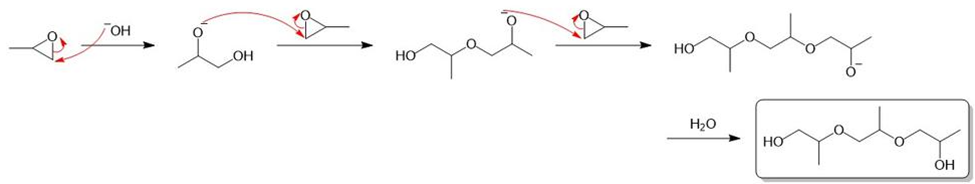
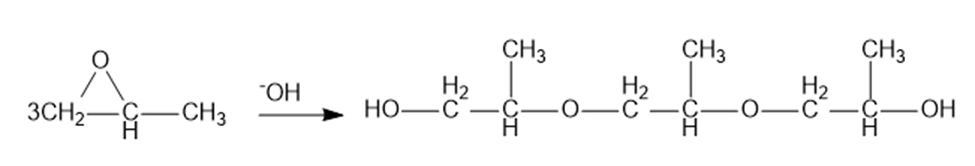
(b) Under base-catalyzed conditions, several molecules of propylene oxide can react to give short polymers. Propose a mechanism for the base-catalyzed formation of the following trimer.
Short Answer
a)

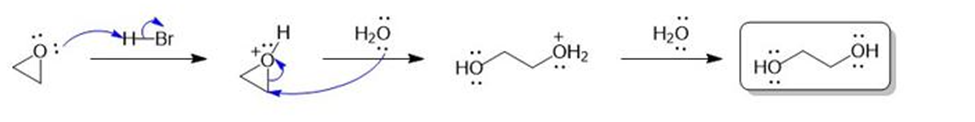
b)