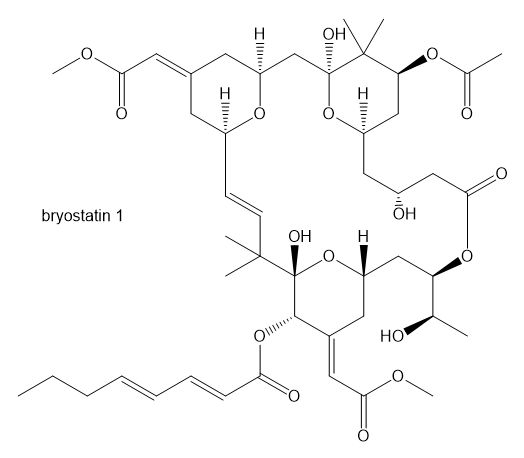
One of the crowning achievements of natural products synthesis was Bryostatin 1, published by Professor Gary Keck (University of Utah; Journal of the American Chemical Society,2011, 133, 744-747). The Bryostatins are a family of compounds isolated from aquatic invertebrates known as Bryozoans. The compounds are of interest for a variety of biological effects, including anti-cancer activity and reversing brain damage in rodents.
a. How many ether functional groups are present in Bryostatin 1?
b. Identify the other oxygen-containing functional groups.
c. This is called a macrolide because it contains a large number of atoms in the large ring. How many atoms are in the large ring?
d. How many chiral centers are in this molecule?
e. Using the number of chiral centers you reported in part (d), calculate the number of stereoisomers possible at these chiral centers. (Ignore stereoisomers at double bonds.)