Chapter 14: Q 51P (page 749)
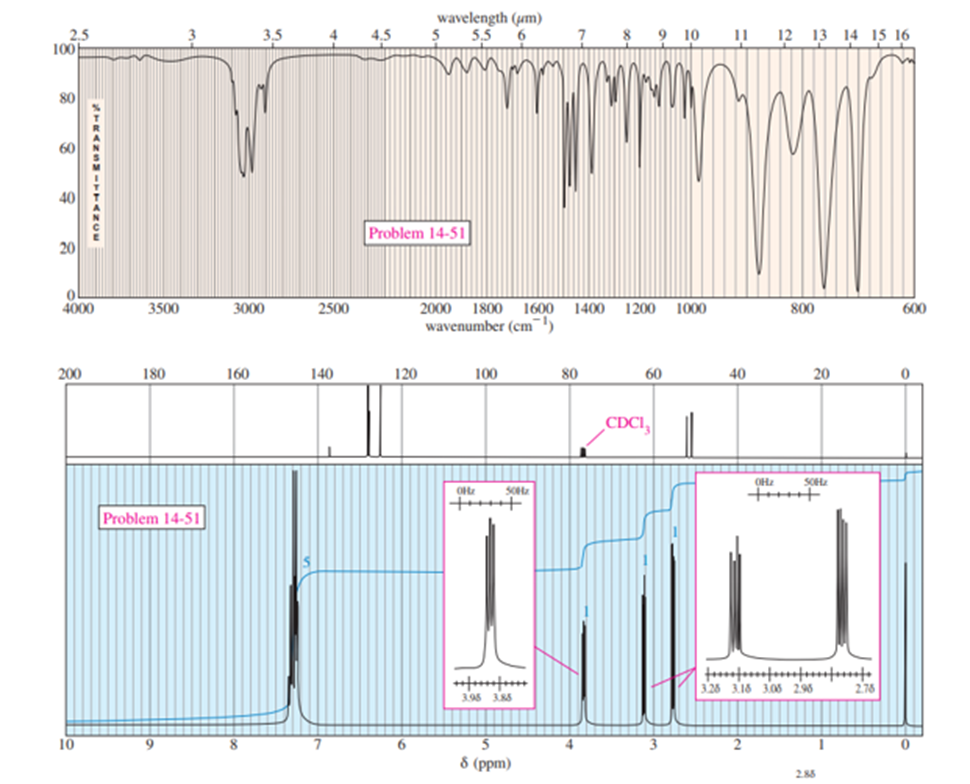
A compound of molecular formula C8H8Ogives the IR and NMR spectra shown here. Propose a structure and show how it is consistent with the observed absorptions.
Short Answer
Learning Materials
Features
Discover
Chapter 14: Q 51P (page 749)
A compound of molecular formula C8H8Ogives the IR and NMR spectra shown here. Propose a structure and show how it is consistent with the observed absorptions.


All the tools & learning materials you need for study success - in one app.
Get started for free
Question. Grignard reactions are often limited by steric hindrance. While Grignard reagents react in high yield with ethylene oxide and monosubstituted epoxides, yields are often lower with disubstituted epoxides. Tri- and tetrasubstituted epoxides react with difficulty, if at all.
(a) Show how to make these alcohols by a Grignard reacting with an epoxide.
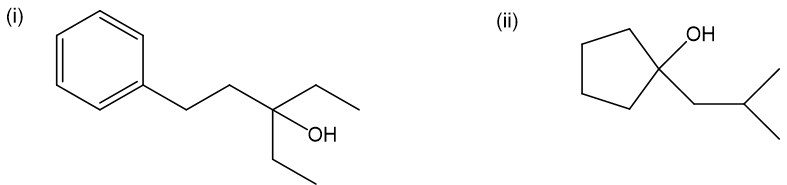
(b) These alcohols cannot be made by a Grignard plus an epoxide. Show the reagents that would be required and why that reaction would be unlikely to succeed.

(a) Tetramethyloxirane is too hindered to undergo nucleophilic substitution by the hindered alkoxide, potassium tert-butoxide. Instead, the product is the allylic alcohol shown. Propose a mechanism to explain this reaction. What type of mechanism does it follow?
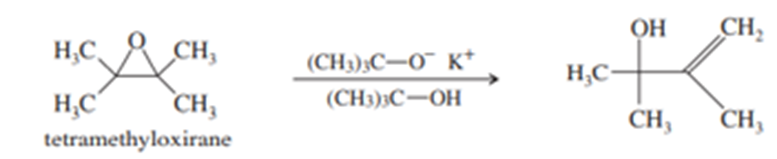
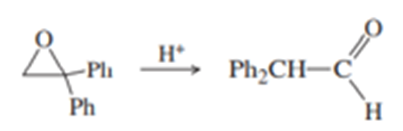
(b) Under mild acid catalysis, 1,1-diphenyloxirane undergoes a smooth conversion to diphenylethanal (diphenylacetaldehyde). Propose a mechanism for this reaction. (Hint: Think Pinacol.)
Question. Glycerol (propane-1,2,3-triol) is a viscous syrup with molecular weight 92g/mol, boiling point 290oC, and density 1.24g/mol. Transforming the three hydroxy groups into their trimethylsilyl ethers (using chlorotrimethylsilane and a tertiary amine) produces a liquid that flows easily, has molecular weight 309g/mol, boiling point approximately 180oC, and density 0.88g/mol. Draw the structures of these two compounds and explain why glycerol has a lower molecular weight but a much higher boiling point and density.
1,4-Dioxane is made commercially by the acid-catalyzed condensation of an alcohol.
(a)Show what alcohol will undergo condensation, with loss of water, to give 1,4-dioxane.
(b)Propose a mechanism for this reaction
(Another true story) An organic lab student carried out the reaction of methyl magnesium iodide with acetone (CH3COCH3), followed by hydrolysis. During the distillation to isolate the product, she forgot to mark the vials she used to collect the fractions. She turned in a product of formula C4H10Othat boiled at 350C.The IR spectrum showed only a weak O-Hstretch around 3300 cm-1, and the mass spectrum showed a base peak at m/z 59.The NMR spectrum showed a quartet (J = 7Hz) of area 3at δ1.3Propose a structure for this product, explain how it corresponds to the observed spectra, and suggest how the student isolated this compound.
What do you think about this solution?
We value your feedback to improve our textbook solutions.