Chapter 14: 14-20P (page 731)
Show how you would accomplish the following transformations. Some of these examples require more than one step.

Short Answer
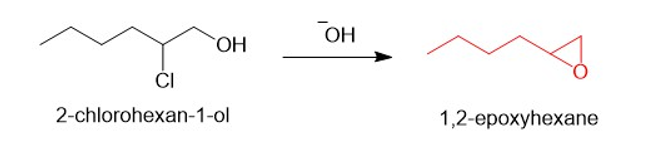
(a)
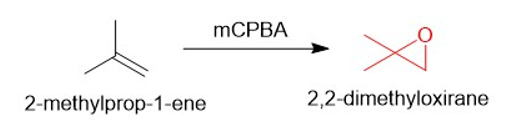
(b)
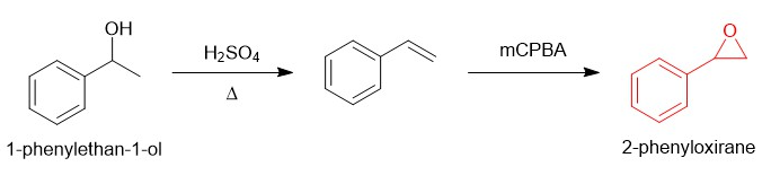
(c)
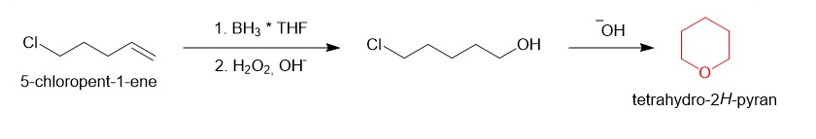
(d)
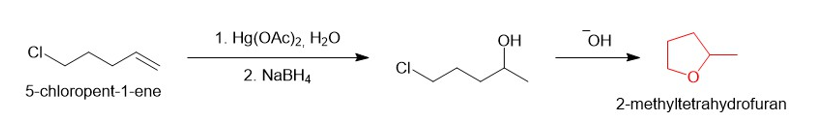
(e)
Learning Materials
Features
Discover
Chapter 14: 14-20P (page 731)
Show how you would accomplish the following transformations. Some of these examples require more than one step.

(a)

(b)

(c)

(d)

(e)

All the tools & learning materials you need for study success - in one app.
Get started for free
Mustard gas, , was used as a poisonous chemical agent in World War I. Mustard gas is much more toxic than a typical primary alkyl chloride. Its toxicity stems from its ability to alkylate amino groups on important metabolic enzymes, rendering the enzymes inactive.
Write structural formulas for the following compounds
(d) diallyl ether (e) allyl ethyl ether (f) cycloheptane oxide
(g) trans-2,3-epoxyheptane (h) (2R,3S)-2-ethoxypentan-3-ol (i) cis-2,3-dimethyloxirane
(Another true story) An organic lab student carried out the reaction of methyl magnesium iodide with acetone (CH3COCH3), followed by hydrolysis. During the distillation to isolate the product, she forgot to mark the vials she used to collect the fractions. She turned in a product of formula C4H10Othat boiled at 350C.The IR spectrum showed only a weak O-Hstretch around 3300 cm-1, and the mass spectrum showed a base peak at m/z 59.The NMR spectrum showed a quartet (J = 7Hz) of area 3at δ1.3Propose a structure for this product, explain how it corresponds to the observed spectra, and suggest how the student isolated this compound.
Show how you would synthesize the following ethers in good yields from the indicated starting materials and any additional reagents needed.
(a) Cyclopentyl n propyl etherfromcyclopentanolandpropan-1-ol.
(b) n-butyl phenyl etherfromphenolandbutan-1-ol.
(c) 2-ethoxyoctanefrom anoctene
(d) 1-methoxydecanefrom adecene
(e) 1-ethoxy-1-methylcyclohexanefrom 2-methylcyclohexanol.
(f) trans-2,3-epoxyoctane from octane-2-ol.
In 2012, a group led by Professor Masayuki Satake of the University of Tokyo reported the isolation and structure determination of a toxin from a marine algal bloom that decimated the fish population off the New Zealand coast in 1998. Extensive mass spectrometry and NMR experiments ultimately led to the structure shown below, named Brevisulcenal-F. (See Journal of the American Chemical Society, 2012, 134, 4963-4968.) This structure holds the record for the largest number of fused rings, at 17.
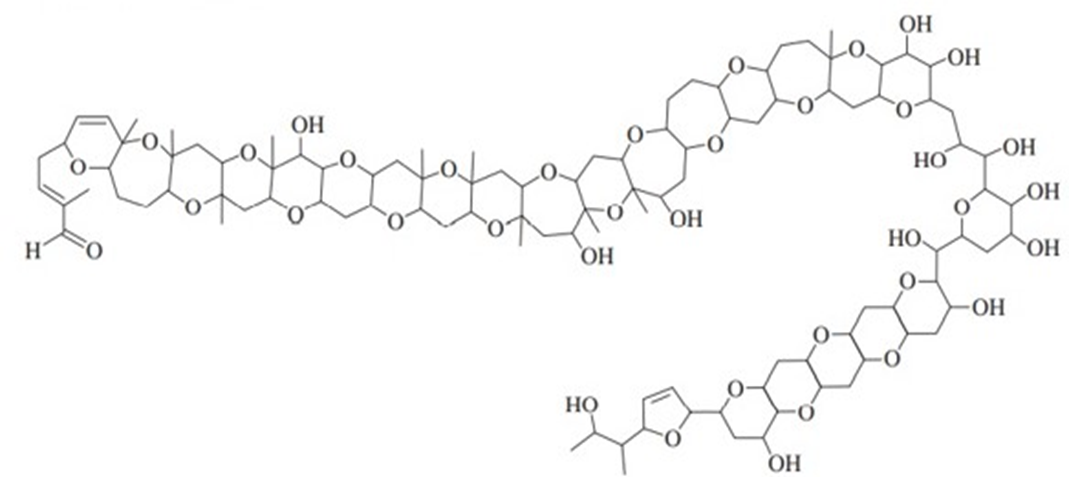
Brevisulcenal-F
a. How many ether groups are present?
b. How many alcohol groups are present? Classify the alcohols as 1o or 2o or 3o.
c. Are there any other oxygen-containing functional groups? Which, if any?
What do you think about this solution?
We value your feedback to improve our textbook solutions.