Chapter 20: 58 P-b (page 1038)
Question: Show the products you expect when each compound reacts with NBS with light shining on the reaction.
b)
Short Answer
b)The product formed from the above compound when react with NBS
Learning Materials
Features
Discover
Chapter 20: 58 P-b (page 1038)
Question: Show the products you expect when each compound reacts with NBS with light shining on the reaction.
b)
b)The product formed from the above compound when react with NBS
All the tools & learning materials you need for study success - in one app.
Get started for free
Show how you would synthesize 2-phenylhex-3-yn-2-ol, starting with acetophenone (PhCOCH3 ) and any other reagents you need. (“2-ol” means there is an OH group on C2.)
Question: Rank the compounds in each set in order of increasing acid strength.
(a),,
(b) ,,
(c)
 ,
, ,
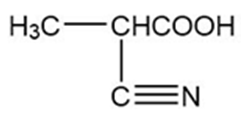
, 
(a) Propose a mechanism for the following reaction: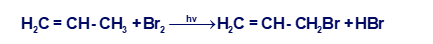
(b) Use the bond-dissociation enthalpies given in Table 4-2 (page 203) to calculate the value of ∆H° for each step shown in your mechanism. (The BDE for is about 280 kJ/mol, or 67 kcal/mol.) Calculate the overall value of ∆H° for the reaction. Are these values consistent with a rapid free-radical chain reaction?
For each compound,
(a)
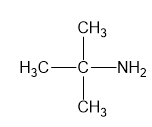
(b)
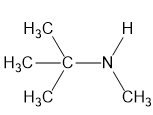
(c)
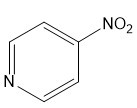
(d)
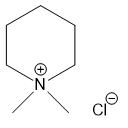
(e)
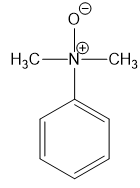
(f)
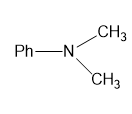
(g)
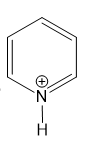
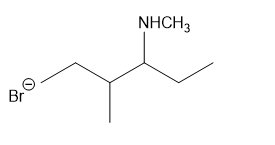
(h)
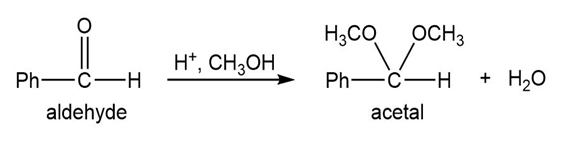
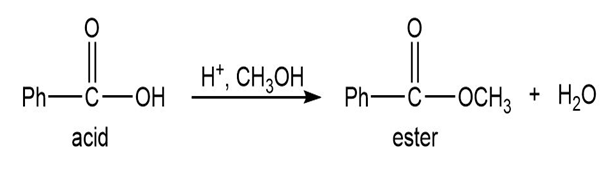
Q.13Most of the Fischer esterification mechanism is identical with the mechanism of acetal formation. The difference is in the final step, where a resonance-stabilized carbocation loses a proton to give the ester. Write mechanisms for the following reactions, with the comparable steps directly above and below each other. Explain why the final step of the esterification (proton loss) cannot occur in acetal formation, and show what happens instead.
What do you think about this solution?
We value your feedback to improve our textbook solutions.