Chapter 20: Problem (page 1038)
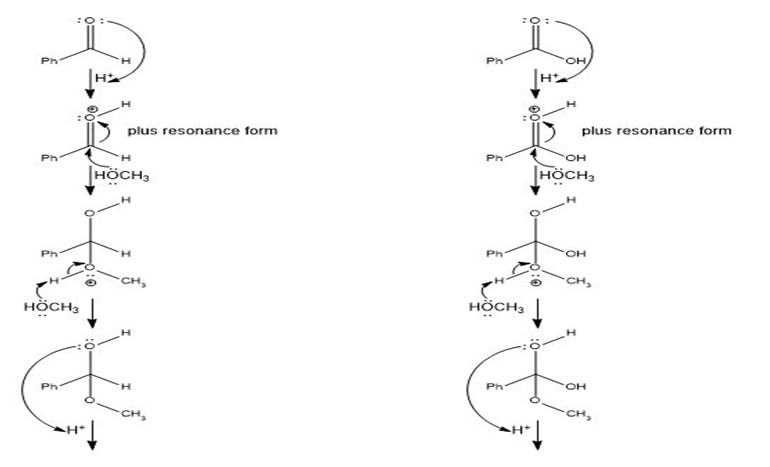
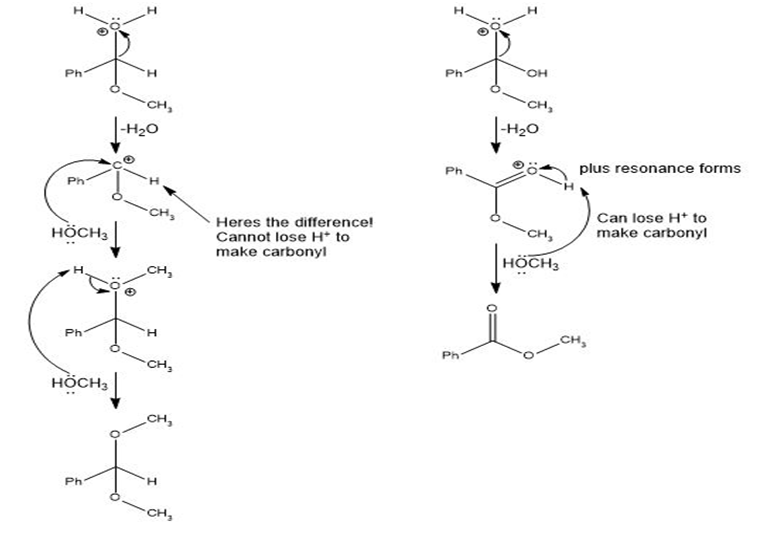
Q.13Most of the Fischer esterification mechanism is identical with the mechanism of acetal formation. The difference is in the final step, where a resonance-stabilized carbocation loses a proton to give the ester. Write mechanisms for the following reactions, with the comparable steps directly above and below each other. Explain why the final step of the esterification (proton loss) cannot occur in acetal formation, and show what happens instead.
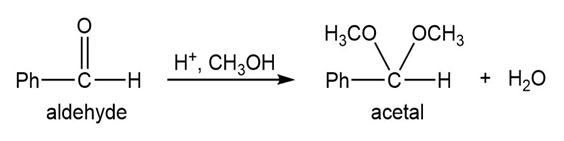
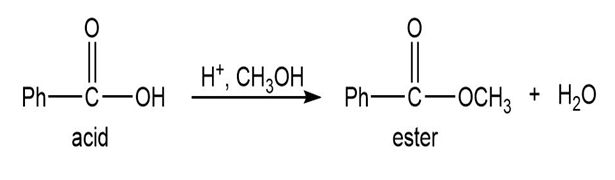
Short Answer
Answer
In the formation of acetal from aldehyde, the loss of H+cannot take place to form carbonyl, and in the formation of ester from acid, the loss ofH+ takes place to form carbonyl.