Chapter 20: Q20P. (page 1067)
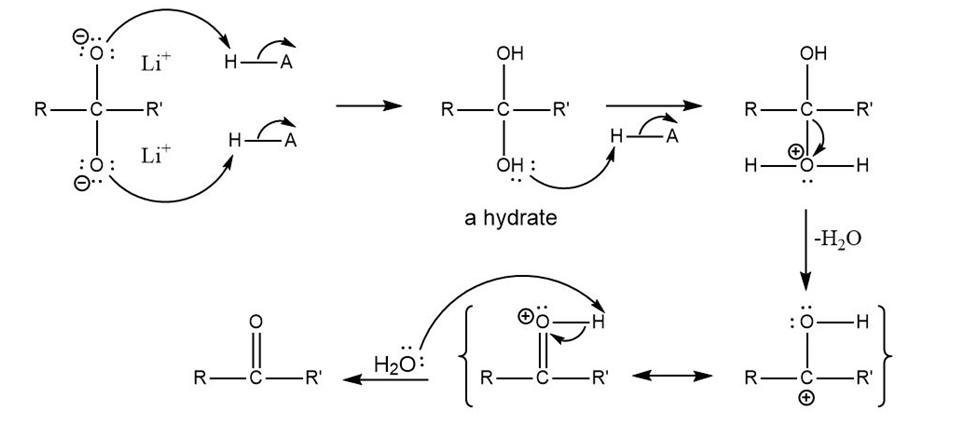
Question: Propose a mechanism for conversion of the dianion to the ketone under mildly acidic conditions.
Short Answer
The mechanism for converting dianion to ketone is as follows:
Learning Materials
Features
Discover
Chapter 20: Q20P. (page 1067)
Question: Propose a mechanism for conversion of the dianion to the ketone under mildly acidic conditions.
The mechanism for converting dianion to ketone is as follows:

All the tools & learning materials you need for study success - in one app.
Get started for free
Question:
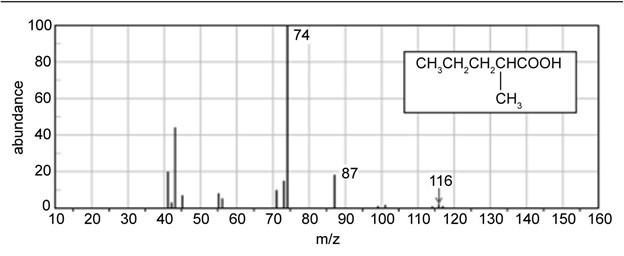
Question: Show the products you expect when each compound reacts with NBS with light shining on the reaction.
a)
Question: What do the following pKa values tell you about the electron-withdrawing abilities of nitro, cyano, chloro, and hydroxy groups?
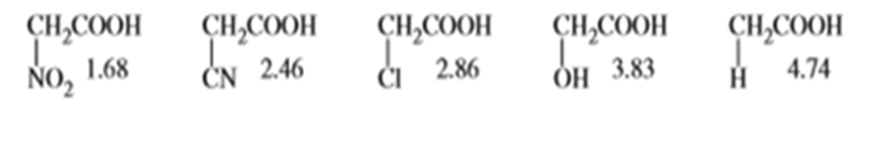
Some of the earliest synthetic detergents were the sodium alkyl sulfates, CH3(CH2)nCH2-OSO3-Na+ .Show how you would make sodium octadecylsulfate using tristearin as your organic starting material.
Predict the major products formed when the following amines undergo exhaustive methylation, treatment with Ag2O, and heating.
5.
6.
What do you think about this solution?
We value your feedback to improve our textbook solutions.