Chapter 20: 58 P-a (page 1038)
Question: Show the products you expect when each compound reacts with NBS with light shining on the reaction.
a)
Short Answer
a)The product formed from the above compound when react with NBS
Learning Materials
Features
Discover
Chapter 20: 58 P-a (page 1038)
Question: Show the products you expect when each compound reacts with NBS with light shining on the reaction.
a)
a)The product formed from the above compound when react with NBS
All the tools & learning materials you need for study success - in one app.
Get started for free
Question. Cellosolve® is the trade name for 2-ethoxyethanol, a common industrial solvent. This compound is produced in chemical plants that use ethylene as their only organic feedstock. Show how you would accomplish this industrial process.
Question: Suppose you have just synthesized heptanoic acid from heptan-1-ol. The product is contaminated by sodium dichromate, sulfuric acid, heptan-1-ol, and possibly heptanal. Explain how you would use acid-base extractions to purify the heptanoic acid. Use a chart, like that in Figure 20-3, to show where the impurities are at each stage.
Predict the products obtained from the reaction of oleic acid with the following reagents.
When pure (S)-lactic acid is esterified by racemic butan-2-ol, the product is 2-butyl lactate, with the following structure:
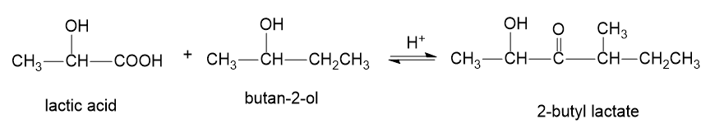
(a)Draw three-dimensional structures of the two stereoisomers formed, specifying the configuration at each asymmetric carbon atom. (Using your models may be helpful.)
(b)Determine the relationship between the two stereoisomers you have drawn.
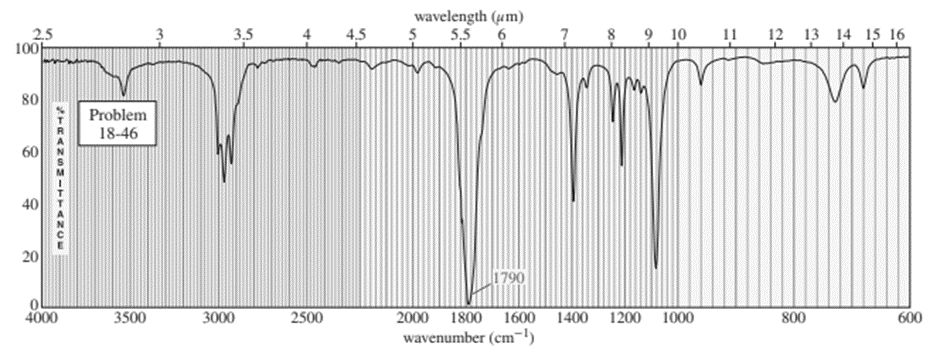
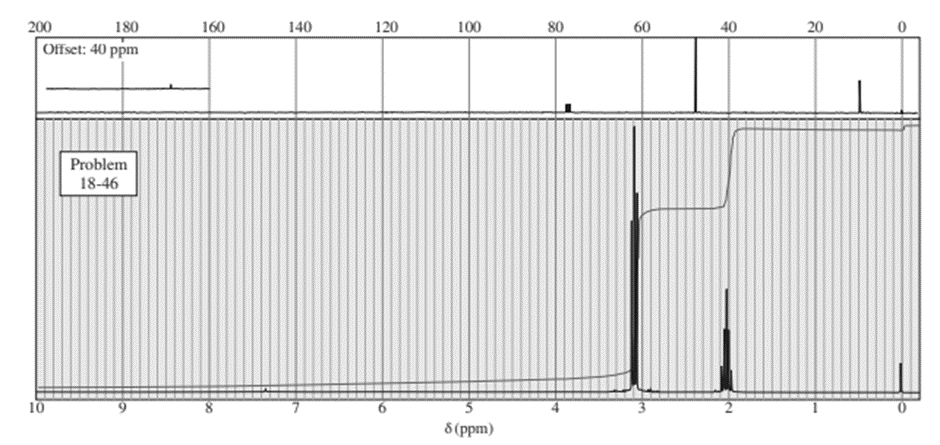
Question:An unknown compound gives a molecular ion of m/z 70 in the mass spectrum. It reacts with semicarbazide hydrochloride to give a crystalline derivative, but it gives a negative Tollens test. The NMR and IR spectra follow. Propose a structure for this compound, and give peak assignments to account for the absorptions in the spectra. Explain why the signal at 1790 cm-1 in the IR spectrum appears at an unusual frequency.
What do you think about this solution?
We value your feedback to improve our textbook solutions.