Acidic hydrolysis of ethylene produces ethanol, and ethylene oxide is produced in large quantities from ethylene and oxygen using a metal catalyst. Peroxyacid can also be used to produce ethylene oxide, but the method is expensive. Ethanol and ethylene oxide react together under acidic conditions to produce cellosolve. Ethylene oxide is a strained molecule. After proton abstraction by oxygen, oxygen acquires a positive charge and strain on the ring further increases; thus, the ring opens up, and carbocation is formed at which ethanol attacks, which leads to the formation of 2-ethoxyethanol.
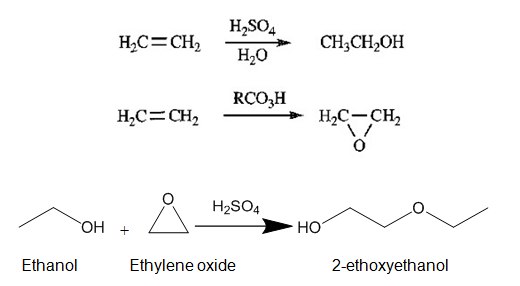
The industrial process of cellosolve













