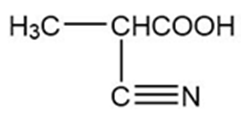
Chapter 20: Q20P (page 1038)
Predict the major products formed when the following amines undergo exhaustive methylation, treatment with Ag2O, and heating.
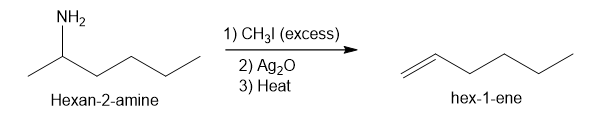
- Hexan-2-amine
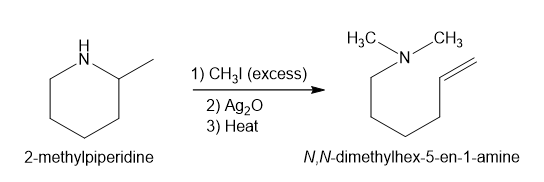
- 2-methylpiperidine
- N-ethylpiperidine
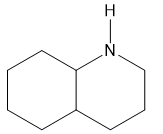
5.
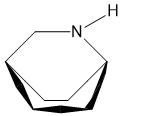
6.
Short Answer
(a)
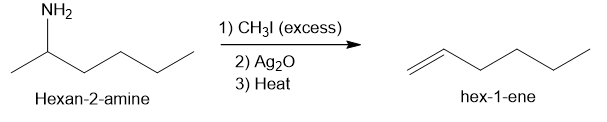
(b)
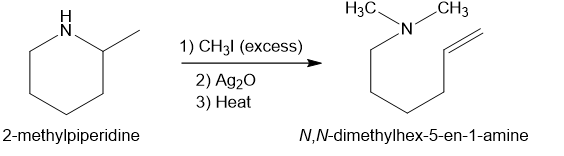
(c)
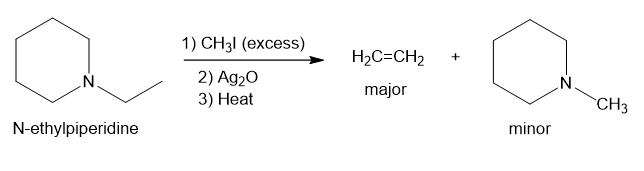
(d)
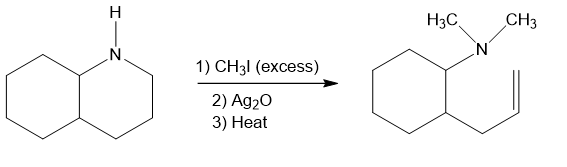
(e)
(f)
Step by step solution
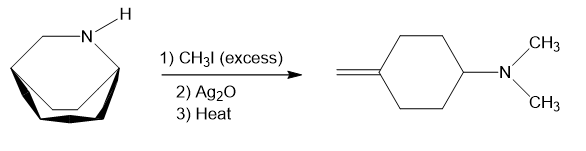
Step-1. Explanation of part (a):
The process of producing tertiary amines and alkenes from quaternary ammonium is called Hofmann elimination. This process can also be referred to as exhaustive methylation. The Hofmann rule states that the major alkene product is the least substituted and least stable product when it comes to asymmetrical amines. Methyl iodide is used in excess because it has no beta hydrogens and therefore cannot compete in an elimination reaction. Silver oxide ion is used for deprotonation of water to form a hydroxide ion.
In part (a), Hofmann exhaustive methylation of the given reactant produces the following product as shown.
Formation of the product
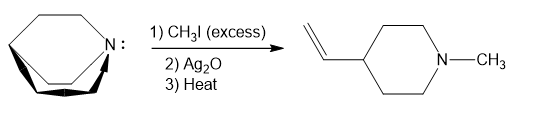
Step-2. Explanation of part (b):
The process of producing tertiary amines and alkenes from quaternary ammonium is called Hofmann elimination. This process can also be referred to as exhaustive methylation. The Hofmann rule states that the major alkene product is the least substituted and least stable product when it comes to asymmetrical amines. Methyl iodide is used in excess because it has no beta hydrogens and therefore cannot compete in an elimination reaction. Silver oxide ion is used for deprotonation of water to form a hydroxide ion.
In part (b), Hofmann exhaustive methylation of the given reactant produces the following product as shown.
Formation of the product
Step-3. Explanation of part (c):
The process of producing tertiary amines and alkenes from quaternary ammonium is called Hofmann elimination. This process can also be referred to as exhaustive methylation. The Hofmann rule states that the major alkene product is the least substituted and least stable product when it comes to asymmetrical amines. Methyl iodide is used in excess because it has no beta hydrogens and therefore cannot compete in an elimination reaction. Silver oxide ion is used for deprotonation of water to form a hydroxide ion.
In part (c), Hofmann exhaustive methylation of the given reactant produces the following product as shown.
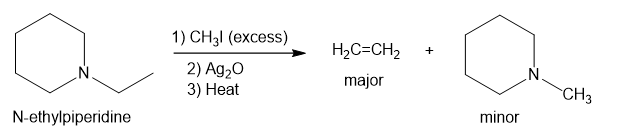
Formation of the product
Step-4. Explanation of part (d):
The process of producing tertiary amines and alkenes from quaternary ammonium is called Hofmann elimination. This process can also be referred to as exhaustive methylation. The Hofmann rule states that the major alkene product is the least substituted and least stable product when it comes to asymmetrical amines. Methyl iodide is used in excess because it has no beta hydrogens and therefore cannot compete in an elimination reaction. Silver oxide ion is used for deprotonation of water to form a hydroxide ion.
In part (d), Hofmann exhaustive methylation of the given reactant produces the following product as shown.
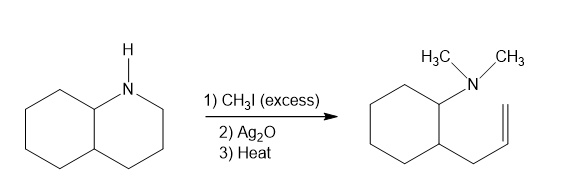
Formation of the product
Step-5. Explanation of part (e):
The process of producing tertiary amines and alkenes from quaternary ammonium is called Hofmann elimination. This process can also be referred to as exhaustive methylation. The Hofmann rule states that the major alkene product is the least substituted and least stable product when it comes to asymmetrical amines. Methyl iodide is used in excess because it has no beta hydrogens and therefore cannot compete in an elimination reaction. Silver oxide ion is used for deprotonation of water to form a hydroxide ion.
In part (e), Hofmann exhaustive methylation of the given reactant produces the following product as shown.
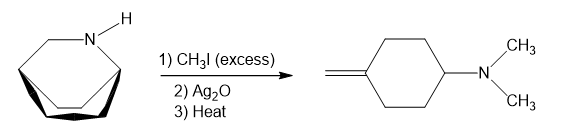
Formation of the product
Step-6. Explanation of part (f):
The process of producing tertiary amines and alkenes from quaternary ammonium is called Hofmann elimination. This process can also be referred to as exhaustive methylation. The Hofmann rule states that the major alkene product is the least substituted and least stable product when it comes to asymmetrical amines. Methyl iodide is used in excess because it has no beta hydrogens and therefore cannot compete in an elimination reaction. Silver oxide ion is used for deprotonation of water to form a hydroxide ion.
In part (f), Hofmann exhaustive methylation of the given reactant produces the following product as shown.
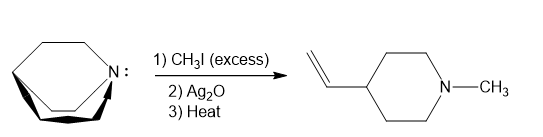
Formation of the product

Unlock Step-by-Step Solutions & Ace Your Exams!
-
Full Textbook Solutions
Get detailed explanations and key concepts
-
Unlimited Al creation
Al flashcards, explanations, exams and more...
-
Ads-free access
To over 500 millions flashcards
-
Money-back guarantee
We refund you if you fail your exam.
Over 30 million students worldwide already upgrade their learning with 91Ӱ��!

 ,
, ,
,