Chapter 20: Q3P. (page 1047)
Question: Rank the compounds in each set in order of increasing acid strength.
(a),,
(b) ,,
(c)
 ,
, ,
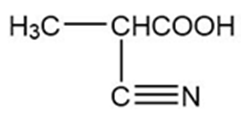
, 
Short Answer
(a)
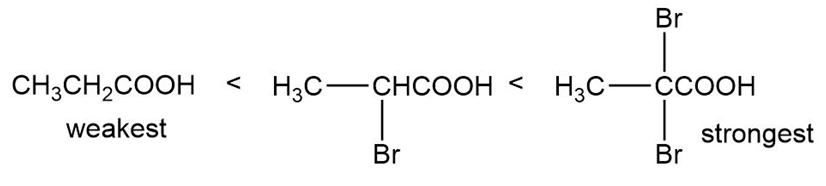
(b)
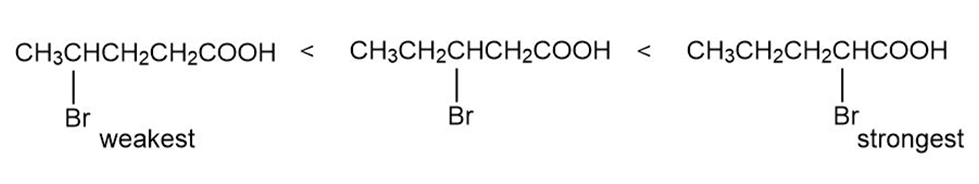
(c)
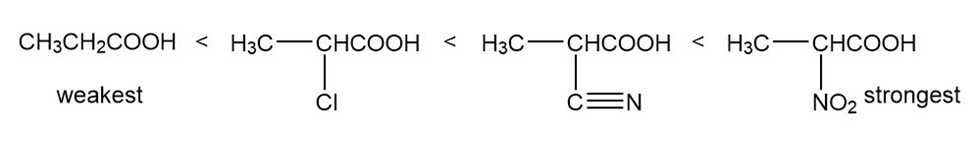
Learning Materials
Features
Discover
Chapter 20: Q3P. (page 1047)
Question: Rank the compounds in each set in order of increasing acid strength.
(a),,
(b) ,,
(c)
 ,
, ,
, 
(a)

(b)

(c)

All the tools & learning materials you need for study success - in one app.
Get started for free
Dimethylamine(CH3) 2 NHhas a molecular weight of 45 and a boiling point of 7.40C. Trimethylamine, (CH3) 3N, has a higher molecular weight (59) but a lower boiling point(3.50C) . Explain this apparent discrepancy.
(a) How many asymmetric carbon atoms are there in an aldotetrose? Draw all the aldotetrose stereoisomers.
(b) How many asymmetric carbon atoms are there in a ketotetrose? Draw all the ketotetrose stereoisomers.
(c) How many asymmetric carbon atoms and stereoisomers are there for an aldohexose? For a ketohexose?
Some of the earliest synthetic detergents were the sodium alkyl sulfates, CH3(CH2)nCH2-OSO3-Na+ .Show how you would make sodium octadecylsulfate using tristearin as your organic starting material.
Question:
Question: A carboxylic acid has two oxygen atoms, each with two nonbonding pairs of electrons.
What do you think about this solution?
We value your feedback to improve our textbook solutions.