Chapter 20: Q10P c. (page 1055)
Question:
- Why do most long-chain fatty acids show a large peak in the mass spectrum at m/z 60 ?
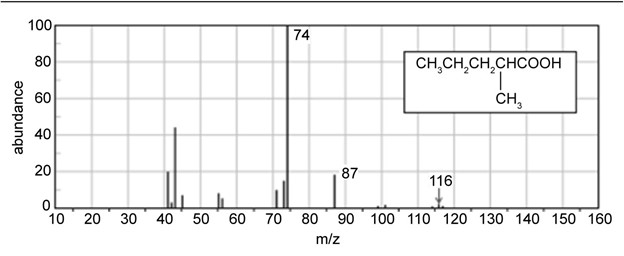
- Use equations to explain the prominent peaks at m/z 74 and m/z 87 in the mass spectrum of 2-methylpentanoic acid.
- Why doesn’t the mass spectrum of 2-methylpentanoic acid show a large peak at m/z 60 ?
Short Answer
Answer
(c) The large peak at m/z 60 shows up if there is a linear carboxylic acid and no substituents present at alpha or beta positions. So, 2-methypentanoic acid has methyl substituent, so it doesn’t show a large peak at m/z 60.