Chapter 20: 58 P-c (page 1038)
Question: Show the products you expect when each compound reacts with NBS with light shining on the reaction.
c)
Short Answer
c)The product formed from the above compound when react with NBS
Learning Materials
Features
Discover
Chapter 20: 58 P-c (page 1038)
Question: Show the products you expect when each compound reacts with NBS with light shining on the reaction.
c)
c)The product formed from the above compound when react with NBS
All the tools & learning materials you need for study success - in one app.
Get started for free
Question: Oxidation of a primary alcohol to an aldehyde usually gives some over-oxidation to the carboxylic acid. Assume you have used PCC to oxidize pentan-1-ol to pentanal.
(b). Which of the expected impurities cannot be removed from pentanal by acid-base extractions? How would you remove this impurity?
Show how the following ketones might be synthesized from the indicated acids, using any necessary reagents.
(a) propiophenone from propionic acid (two ways, using alkylation of the acid and using Friedel-Crafts acylation)
(b) methyl cyclohexyl ketone from cyclohexanecarboxylic acid
Which of the following compounds are chiral? Draw each compound in its most symmetric conformation, star any asymmetric carbon atoms, and draw any mirror planes. Label any meso compounds. You may use Fischer projections if you prefer.
(a) meso-2,3-dibromo-2,3-dichlorobutane
(b) -2,3-dibromo-2,3-dichlorobutane
(c) (2R,3S)-2-bromo-3-chlorobutane
(d) (2R,3S)-2,3-dibromobutane
(e) (R,R)-2,3-dibromobutane
(f)
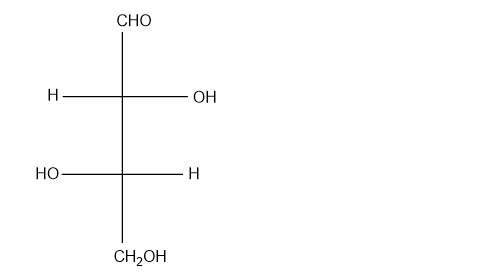
(g)
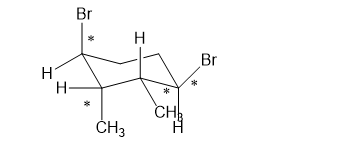
(h)

Question: Draw all four resonance forms of the fragment at m/z 73 in the mass spectrum of pentanoic acid.
(a)Hydrogen peroxide (HOOH) has a pKa of 11.6, making it roughly 10,000 times as strong an acid as water (pKa = 15.7). Explain why H2O2is a stronger acid than H2O.
(b) In contrast to part (a), peroxyacetic acid (pKa = 8.2) is a much weakeracid than acetic acid (pka = 4.74) . Explain why peroxyacetic acid is a weaker acid than acetic acid.
(c) Peroxyacetic acid (bp = 105° C) has a lower boiling point than acetic acid (bp = 118° C) , even though peroxyacetic acid has a higher molecular weight. Explain why peroxyacetic acid is more volatile than acetic acid.
What do you think about this solution?
We value your feedback to improve our textbook solutions.