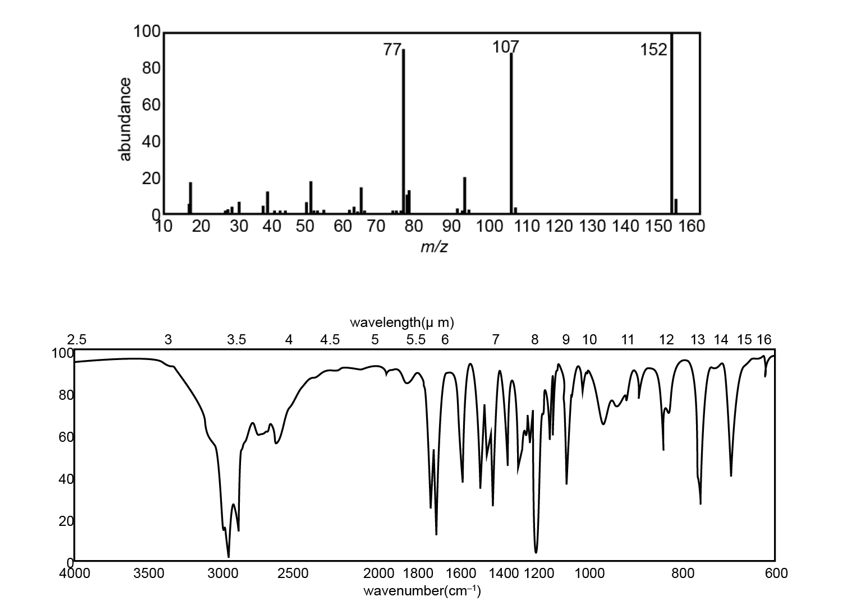
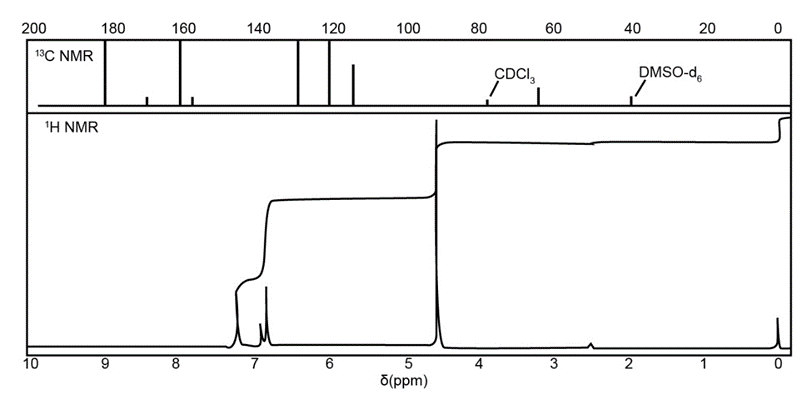
Chapter 20: Q-5-21P (page 1038)
Which of the following compounds are chiral? Draw each compound in its most symmetric conformation, star any asymmetric carbon atoms, and draw any mirror planes. Label any meso compounds. You may use Fischer projections if you prefer.
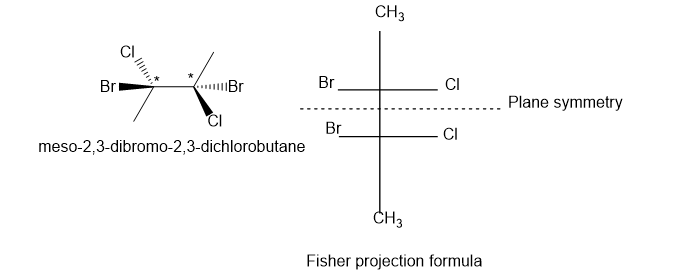
(a) meso-2,3-dibromo-2,3-dichlorobutane
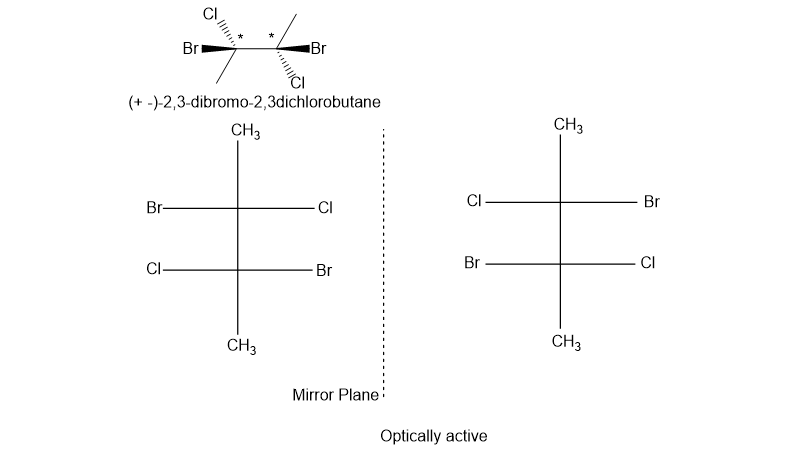
(b) -2,3-dibromo-2,3-dichlorobutane
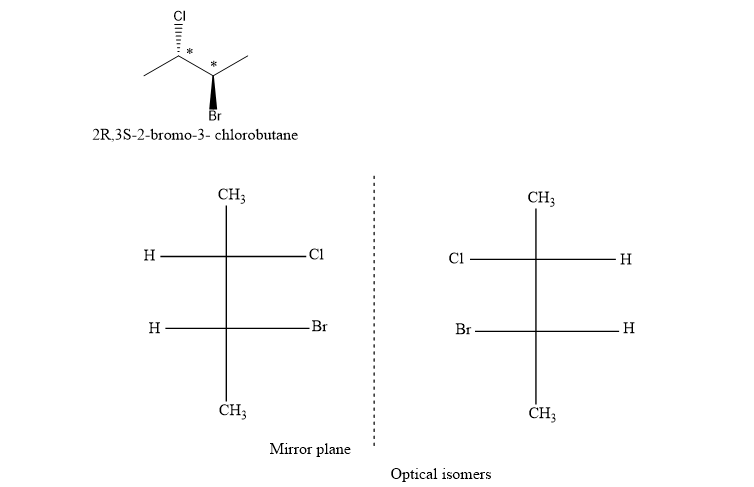
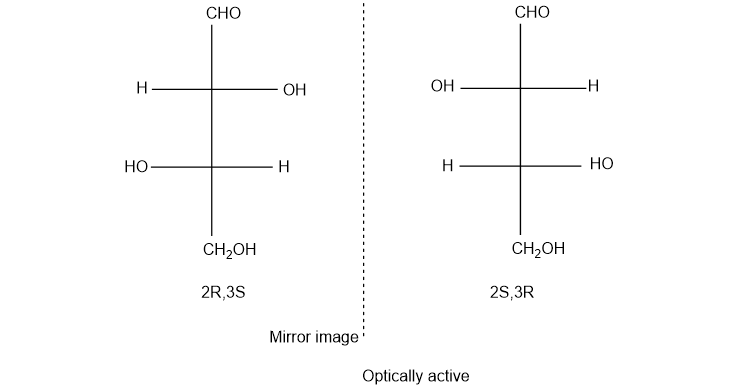
(c) (2R,3S)-2-bromo-3-chlorobutane
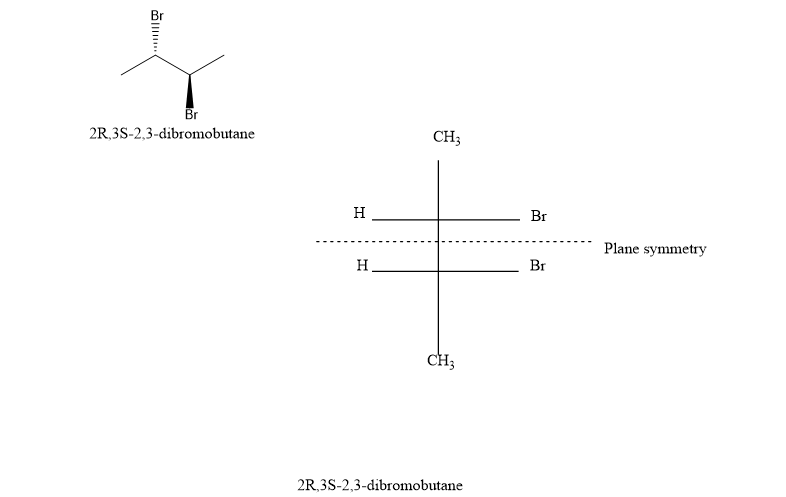
(d) (2R,3S)-2,3-dibromobutane
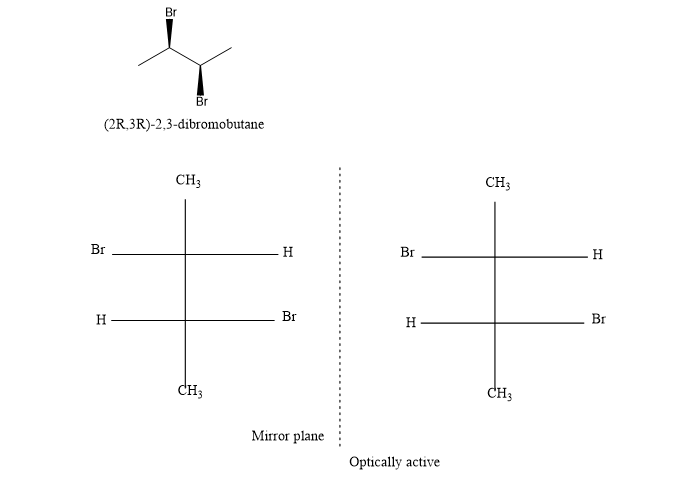
(e) (R,R)-2,3-dibromobutane
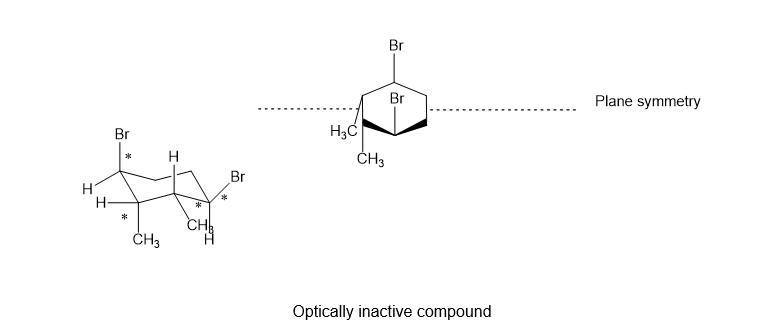
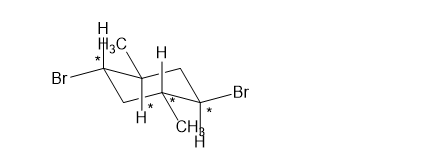
(f)
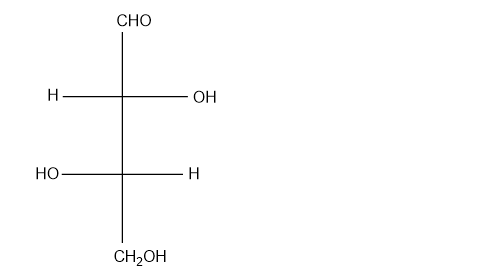
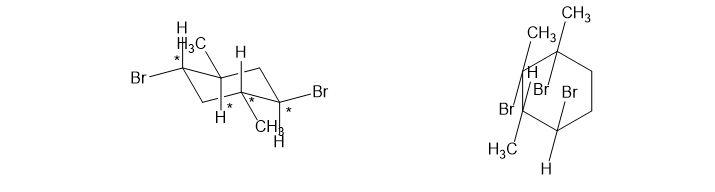
(g)
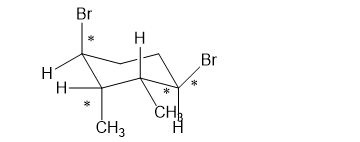
(h)
Short Answer
(a). It is not a chiral compound.
(b). It is a chiral compound.
(c). It is a chiral compound.
(d). It is not a chiral compound.
(e). It is a chiral compound.
(f). It is a chiral compound.
(g). It is not a chirtal compound.
(h). It is a chiral compound.