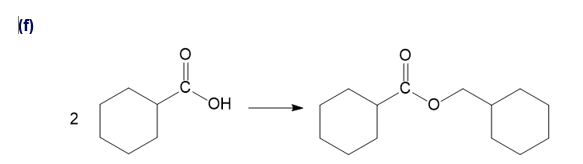
Chapter 20: Q51P (page 1038)
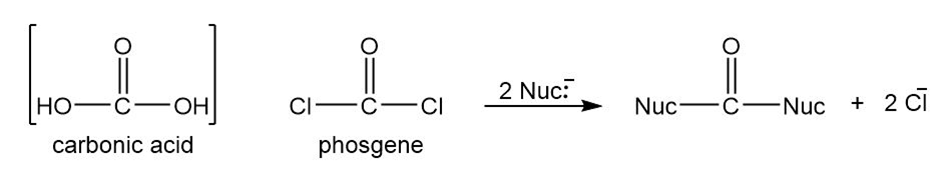
Phosgene is the acid chloride of carbonic acid. Although phosgene was used as a war gas in World War I, it is now used as a reagent for the synthesis of many useful products. Phosgene reacts like other acid chlorides, but it can react twice.
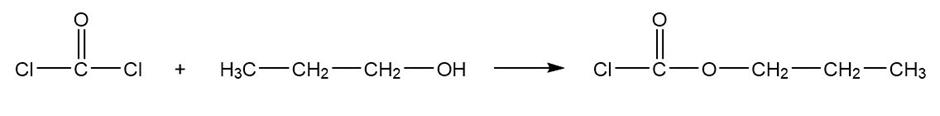
(a) Predict the products formed when phosgene reacts with excess propan-1-ol.
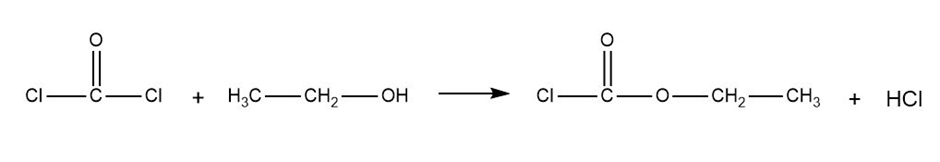
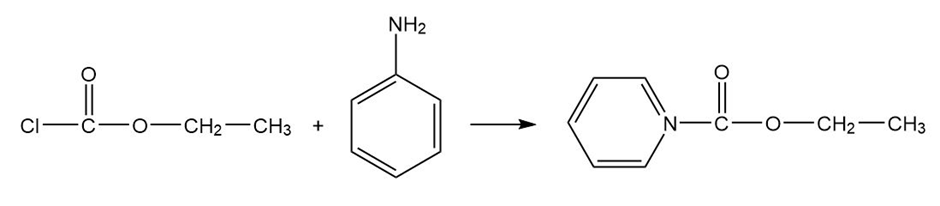
(b) Predict the products formed when phosgene reacts with 1 equivalent of ethanol, followed by 1 equivalent of aniline.
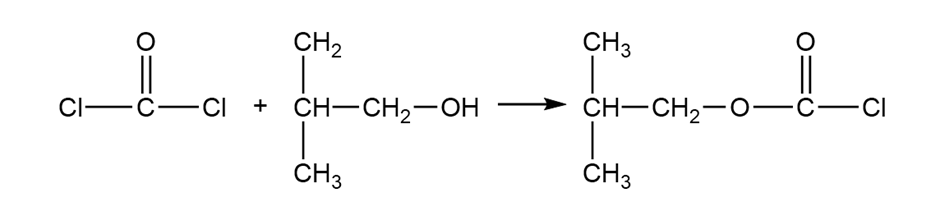
(c) Iso-butyloxycarbonyl chloride is an important reagent for the synthesis of peptides and proteins. Show how you would use phosgene to synthesize iso-butyloxycarbonyl chloride.
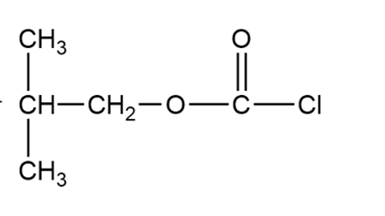
Isobutyloxycarbonyl chloride
Short Answer
(a)
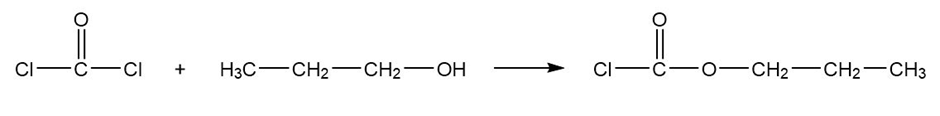
Phosgene propan-1-ol propyl carbonochloridate
Formation of propyl carbonochloridate
(b)

Phosgene Ethanol Ethyl carbonochloridate
Formation of ethyl carbonochloridate

Ethyl carbonochloridate Aniline
Formation of the desired product
(c)
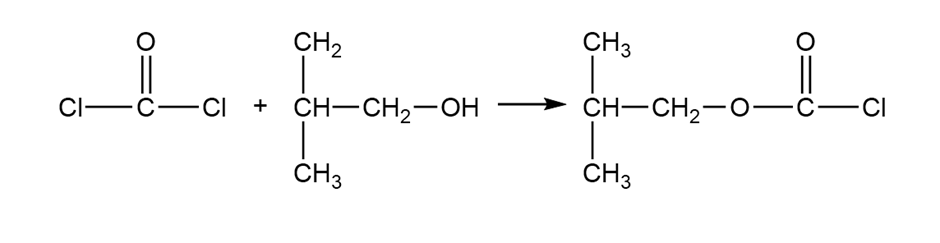
Formation of isobutyloxycarbonyl chloride



 ,
, ,
,