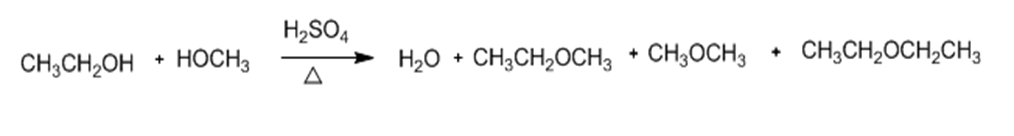Chapter 20: Q25P (page 1038)
Explain why the acid-catalyzed condensation is a poor method for the synthesis of an unsymmetrical ether such as ethyl methyl ether, CH3CH2 - O - CH3.
Short Answer
The acid-catalyzed condensation is a poor method for synthesizing unsymmetrical ethers as the unsymmetrical alcohols generate a mixture of products.