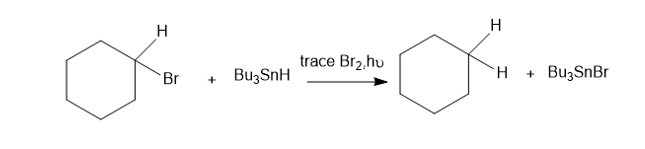
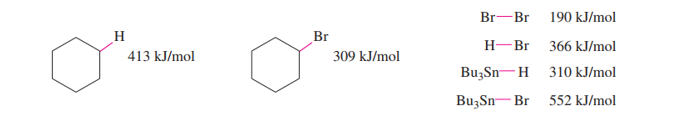
Chapter 20: Q24P (page 1038)
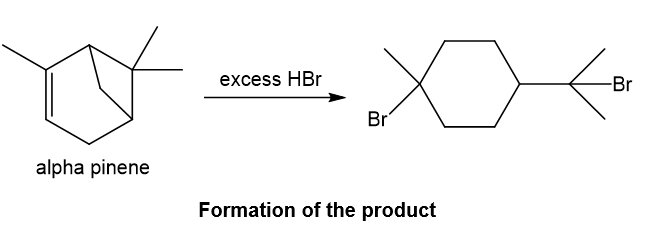
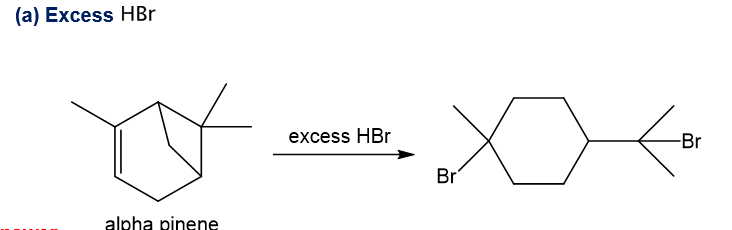
The structure of alpha-pinene appears in Figure 25-1. Predict the products formed when alpha-pinene reacts with the following reagents.
a. Excess HBr
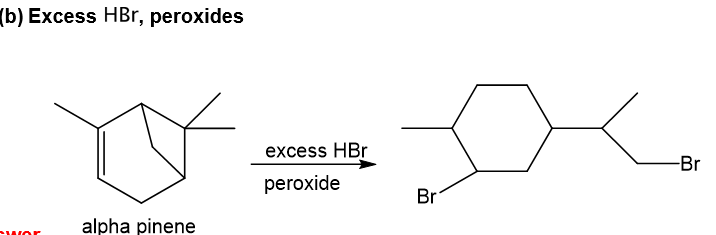
b.Excess HBr, peroxides
c. Excess Br2 in CCl4
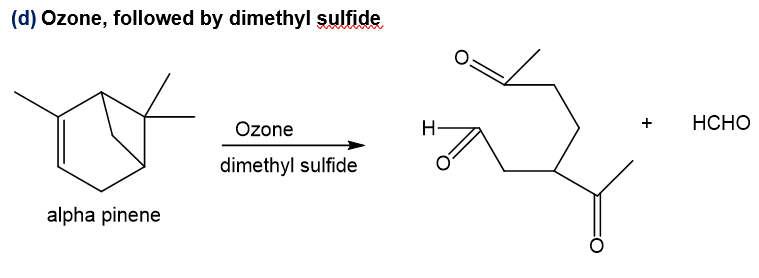
d.Ozone, followed by dimethyl sulfide
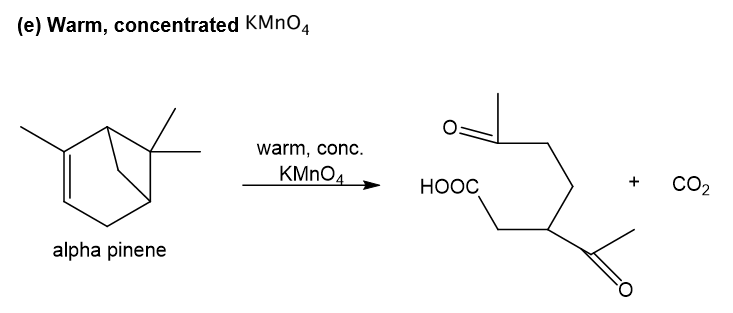
e. Warm, concentrated KMnO4
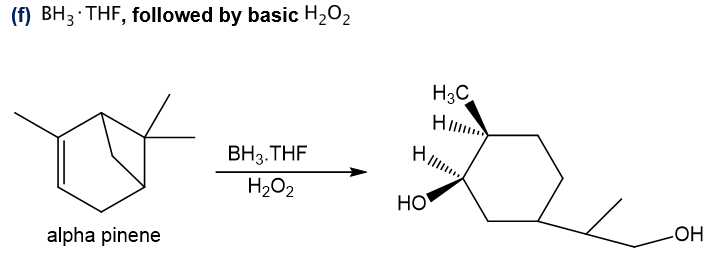
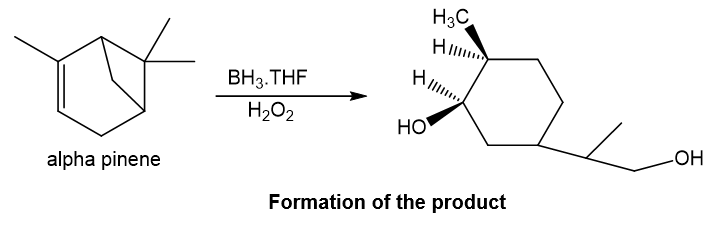
f.BH3 , THF followed by basic H2O2
Short Answer
Alpha-pinene undergoes reaction with excess hydrogen bromide and forms the product as depicted. With hydrogen bromide in absence of peroxide, Markovnikov addition takes place in which bromine gets attached to the more substituted carbon of the double bond and also the four-membered ring in the structure opens up in presence of proton of hydrogen bromide as four-membered ring is strained, then bromine also gets attached to the carbocation which is formed after the ring opening and required product is formed.

Step by step solution
Step-1. Explanation of part (a):
Alpha-pinene undergoes reaction with excess hydrogen bromide and forms the product as depicted. With hydrogen bromide in absence of peroxide, Markovnikov addition takes place in which bromine gets attached to the more substituted carbon of the double bond and also the four-membered ring in the structure opens up in presence of proton of hydrogen bromide as four-membered ring is strained, then bromine also gets attached to the carbocation which is formed after the ring opening and required product is formed.

Step-2. Explanation of part (b):
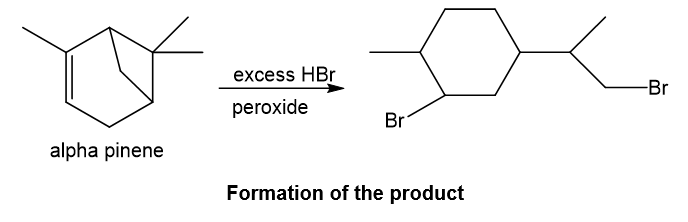
Alpha-pinene undergoes reaction with excess hydrogen bromide and forms the product as depicted. With hydrogen bromide in presence of peroxide, anti-Markovnikov addition takes place in which bromine gets attached to the less substituted carbon of the double bond and also the four-membered ring in the structure opens up in presence of proton of hydrogen bromide as four-membered ring is strained, then bromine also gets attached to the carbocation which is formed after the ring opening and attaches to less substituted carbon and required product is formed.
Step-3. Explanation of part (c):
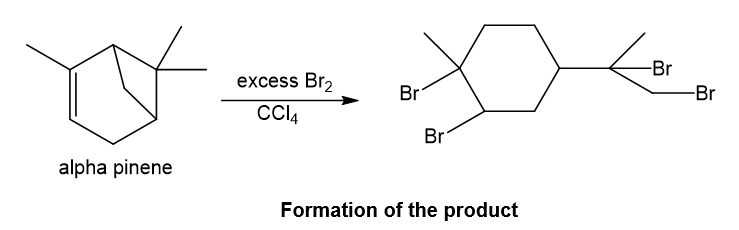
Alpha-pinene undergoes reaction with excess bromine in presence of carbon tetrachloride which is a solvent and forms the product as depicted. With bromine molecule, addition reaction of bromine takes place in which bromine gets attached to the more and less substituted carbons of the double bond and also the four-membered ring in the structure opens up in presence of proton of hydrogen bromide as four-membered ring is strained, then bromine also gets attached to the carbocation which is formed after the ring opening and required product is formed.
Step-4. Explanation of part (d):
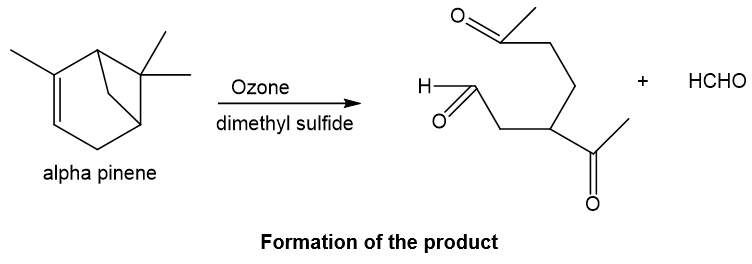
Alpha-pinene on reaction with ozone followed by dimethyl sulfide, undergoes reductive ozonolysis reaction in which the oxygen gets added to both ends of the double bond and aldehyde and ketone gets formed. Also, ketone formation occurs at the other double bond which is formed after ring opening of four-membered ring which leads to carbocation and eventually double bond formation after deprotonation and at this double bond, ketone formation occurs when oxygen is added, and formaldehyde is the side product.
Step-5. Explanation of part (e):
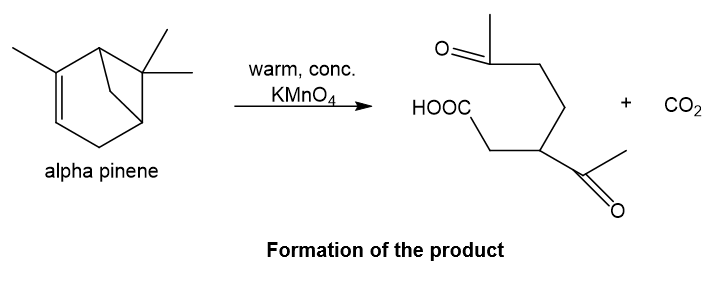
Alpha-pinene on reaction with warm, concentrated potassium permanganate undergoes oxidation reaction at the double bond in which oxygen is added at the ends of double bond and also over-oxidation occurs as potassium permanganate is strong oxidising agent, thus aldehydic group gets converted to carboxylic acid group. Also, ketone formation occurs at the other double bond which is formed after ring opening of four-membered ring which leads to carbocation and eventually double bond formation after deprotonation and at this double bond, ketone formation occurs when oxygen is added and carbon dioxide gas is the side product.
Step-6. Explanation of part (f):
Alpha-pinene undergoes hydroboration-oxidation reaction in presence of boron hydride and hydrogen peroxide. Substitution of hydroxyl group occurs at the less substituted carbon of the double bond in the final product. Also, the substitution of hydroxyl group occurs at the other double bond’s less substituted carbon which is formed after the ring-opening reaction of four-membered ring as it is strained.

Unlock Step-by-Step Solutions & Ace Your Exams!
-
Full Textbook Solutions
Get detailed explanations and key concepts
-
Unlimited Al creation
Al flashcards, explanations, exams and more...
-
Ads-free access
To over 500 millions flashcards
-
Money-back guarantee
We refund you if you fail your exam.
Over 30 million students worldwide already upgrade their learning with 91Ӱ��!