Chapter 20: Q61P (page 1038)
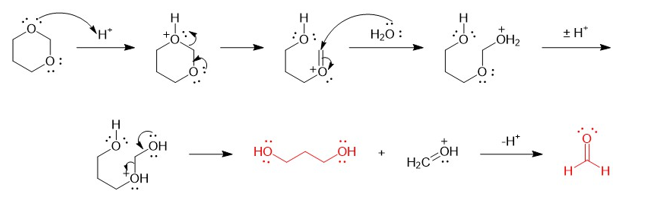
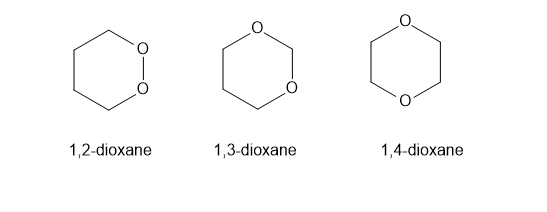
There are three dioxane isomers: 1,2-dioxane, 1,3- dioxane, and 1,4-dioxane. One of these acts like an ether and is an excellent solven for Grignard reactions. Another one is potentially explosive when heated. The third one quickly hydrolyzes in dilute acid. Show which isomer acts like a simple ether, and then explain why one of them is potentially explosive. Propose a mechanism for the acid hydrolysis of the third isomer.
Short Answer
behaves like a simple ether. easily hydrolyzes in aqueous acid. is potentially explosive.
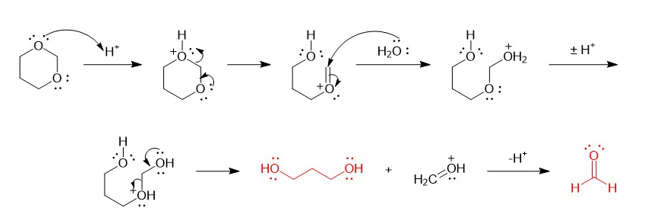
Mechanism for acidic hydrolysis of 1, 3 dioxanes