In the IR spectrum, the sharp spike at 2250 cm-1 corresponds to C≡N . The peak at 1750 cm-1 corresponds to C=O, and the peak at 1200 cm-1 corresponds to C-O. The C=O and C-O peaks indicate that the compound may be an ester.
In the NMR spectrum, the triplet and quartet indicate the presence of -CH3CH2 group. The quartet at δ =4.3 corresponds to (CH3CH2O-) group. The 2H singlet at δ =3.5 corresponds to an isolated CH2.
The fragments can be combined in two ways:
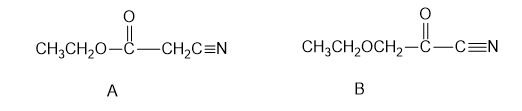
Structures generated by combining the fragments
The NMR proves the structure is A. If the structure is B, the CH2 between oxygen and the carbonyl will come farther downfield than the CH2 of the ethyl, which is only deshielded by the oxygen atom. This does not happen, so the correct structure is A.
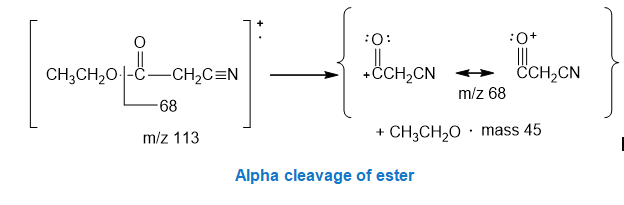
The peak in the mass spectrum at m/z 68 is due to the alpha cleavage of the ester