Chapter 20: Q19c (page 1066)
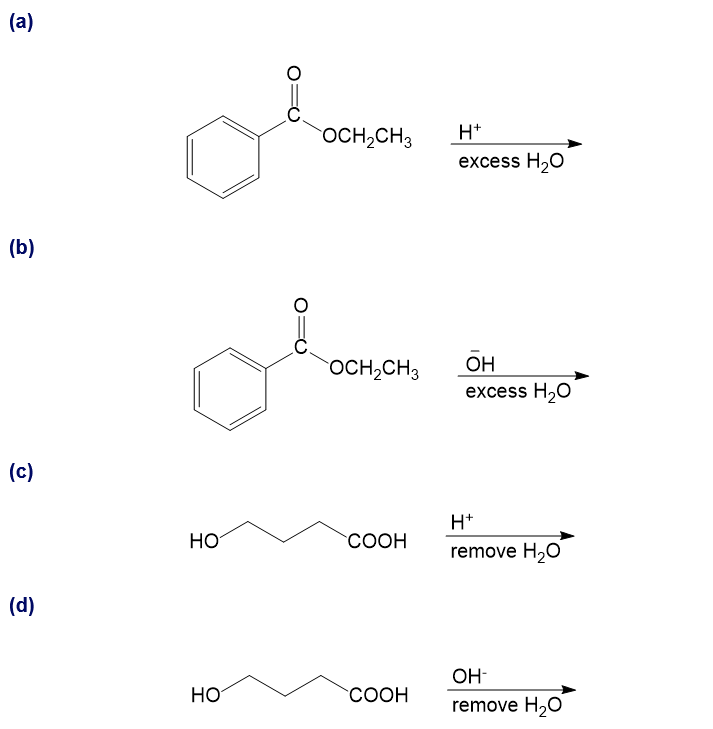
Show how you would synthesize the following compounds from the appropriate carboxylic acids or acid derivatives.
a)
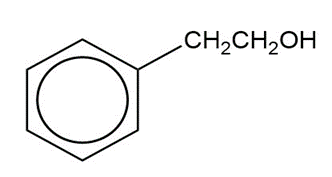
b)
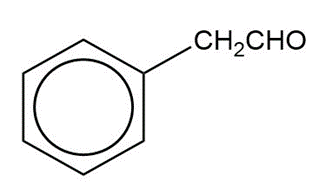
c)
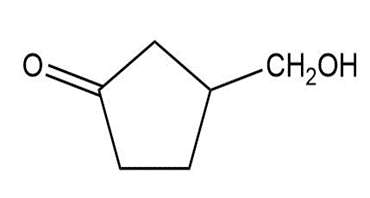
Short Answer
c)
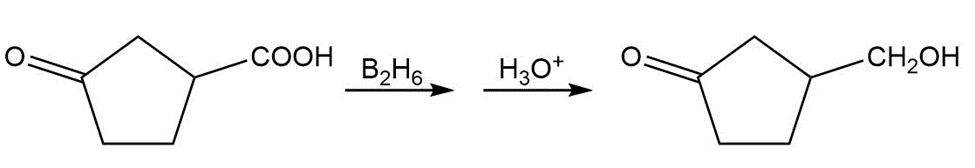
Learning Materials
Features
Discover
Chapter 20: Q19c (page 1066)
Show how you would synthesize the following compounds from the appropriate carboxylic acids or acid derivatives.
a)

b)

c)

c)

All the tools & learning materials you need for study success - in one app.
Get started for free
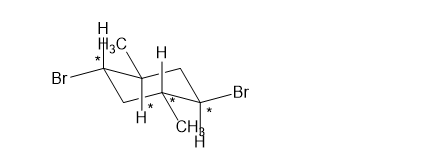
Which of the following compounds are chiral? Draw each compound in its most symmetric conformation, star any asymmetric carbon atoms, and draw any mirror planes. Label any meso compounds. You may use Fischer projections if you prefer.
(a) meso-2,3-dibromo-2,3-dichlorobutane
(b) -2,3-dibromo-2,3-dichlorobutane
(c) (2R,3S)-2-bromo-3-chlorobutane
(d) (2R,3S)-2,3-dibromobutane
(e) (R,R)-2,3-dibromobutane
(f)
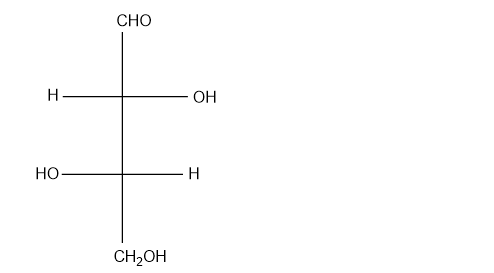
(g)
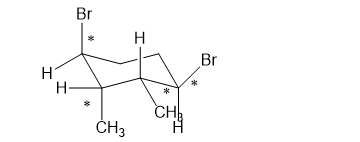
(h)
Question: What do the following pKa values tell you about the electron-withdrawing abilities of nitro, cyano, chloro, and hydroxy groups?
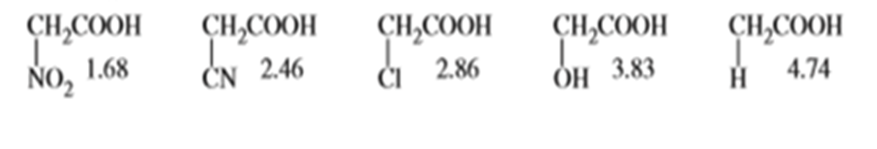
Dimethylamine(CH3) 2 NHhas a molecular weight of 45 and a boiling point of 7.40C. Trimethylamine, (CH3) 3N, has a higher molecular weight (59) but a lower boiling point(3.50C) . Explain this apparent discrepancy.
Show how you would accomplish the following syntheses efficiently (you may use any necessary reagents).
(a)trans-1-�����dz��Dz��ܳ�-2-��Ա�→trans-pent-3-enoic acid (two ways)
(b)hex-3-ene→propanoic acid
(c) but-2-enal→but-2-enoic acid
(d) hexanoic acid→hexanal
(e)
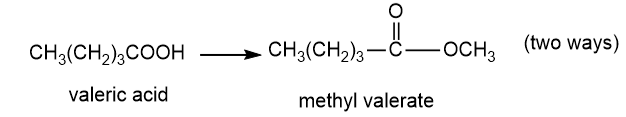
(f)
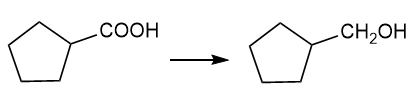
(g)
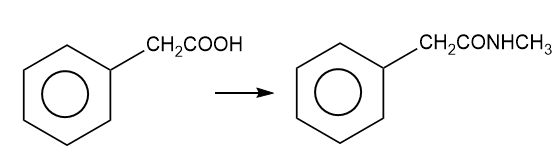
(h)
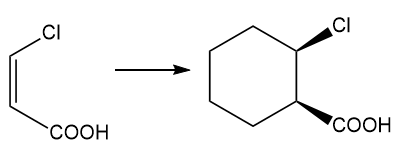
Predict the productsand propose mechanisms for the following reactions.
What do you think about this solution?
We value your feedback to improve our textbook solutions.