Chapter 20: Q18P c. (page 1065)
Show how to synthesize the following compounds, using appropriate carboxylic acids and amines.
(a)
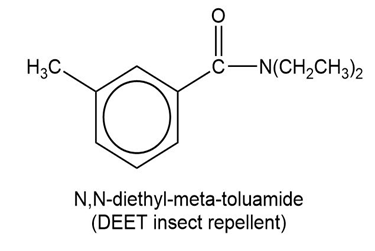
(b)
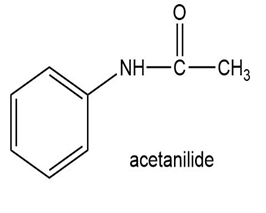
(c)
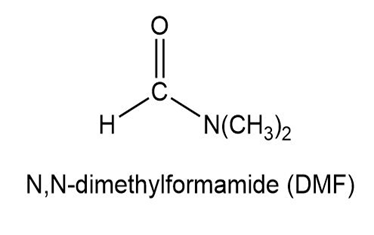
Short Answer
(c)
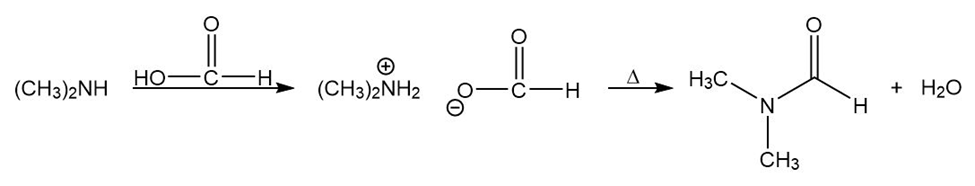
Learning Materials
Features
Discover
Chapter 20: Q18P c. (page 1065)
Show how to synthesize the following compounds, using appropriate carboxylic acids and amines.
(a)

(b)

(c)

(c)

All the tools & learning materials you need for study success - in one app.
Get started for free
Show how you would use an acid chloride as an intermediate to synthesize(a) N-phenylbenzamide from benzoic acid and aniline.(b) phenyl propionate from propionic acid and phenol.
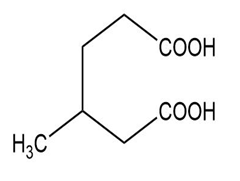
Question: Name the following carboxylic acids (when possible, give both a common name and a systematic name).
(a)
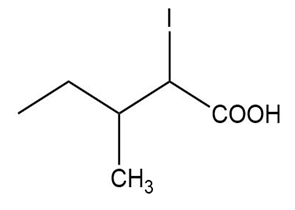
(b)
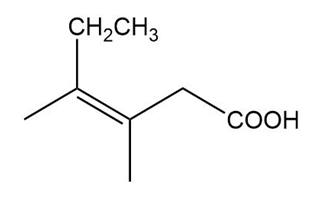
(c)
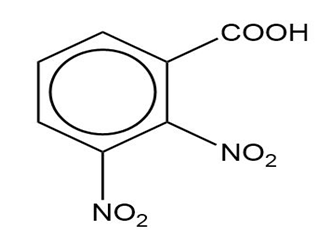
(d)
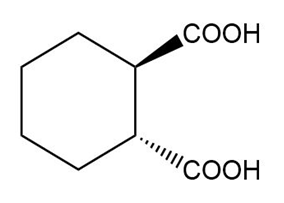
(e)
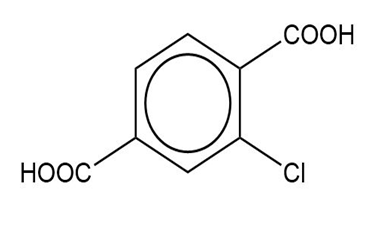
(f)
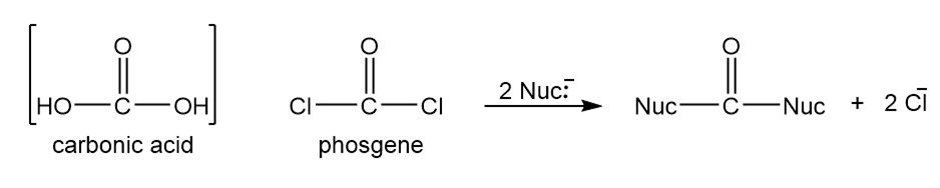
Phosgene is the acid chloride of carbonic acid. Although phosgene was used as a war gas in World War I, it is now used as a reagent for the synthesis of many useful products. Phosgene reacts like other acid chlorides, but it can react twice.
(a) Predict the products formed when phosgene reacts with excess propan-1-ol.
(b) Predict the products formed when phosgene reacts with 1 equivalent of ethanol, followed by 1 equivalent of aniline.
(c) Iso-butyloxycarbonyl chloride is an important reagent for the synthesis of peptides and proteins. Show how you would use phosgene to synthesize iso-butyloxycarbonyl chloride.
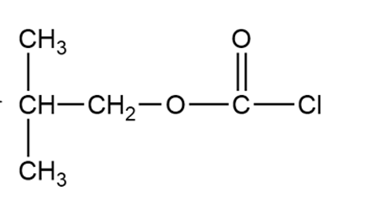
Isobutyloxycarbonyl chloride
Show how Fischer esterification might be used to form the following esters. In each case, suggest a method for driving the reaction to completion.
(a) methyl salicylate
(b) methyl formate (bp 32C)
(c) ethyl phenylacetate
Question: Phenols are less acidic than carboxylic acids, with values of around 10. Phenols are deprotonated by (and therefore soluble in) solutions of sodium hydroxide but not by solutions of sodium bicarbonate. Explain how you would use extractions to isolate the three pure compounds from a mixture of p-cresol (p-methylphenol), cyclohexanone, and benzoic acid.
What do you think about this solution?
We value your feedback to improve our textbook solutions.