Chapter 20: Q18P b. (page 1065)
Show how to synthesize the following compounds, using appropriate carboxylic acids and amines.
(a)
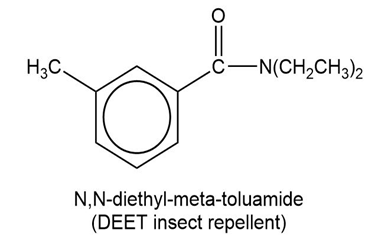
(b)
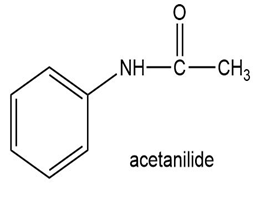
(c)
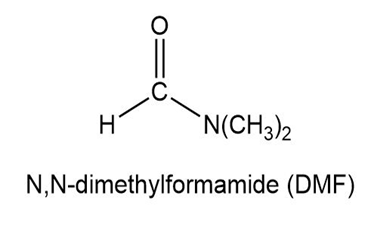
Short Answer
(b)
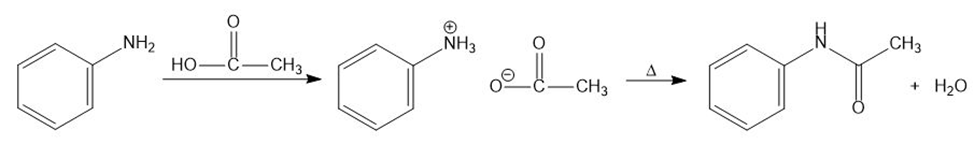
Learning Materials
Features
Discover
Chapter 20: Q18P b. (page 1065)
Show how to synthesize the following compounds, using appropriate carboxylic acids and amines.
(a)

(b)

(c)

(b)

All the tools & learning materials you need for study success - in one app.
Get started for free
(a) Give the products expected when acetic formic anhydride reacts with
(i) aniline and
(ii) benzyl alcohol
(b) Propose mechanisms for these reactions.
Question: A carboxylic acid has two oxygen atoms, each with two nonbonding pairs of electrons.
The IR, NMR, and mass spectra are provided for an organic compound.
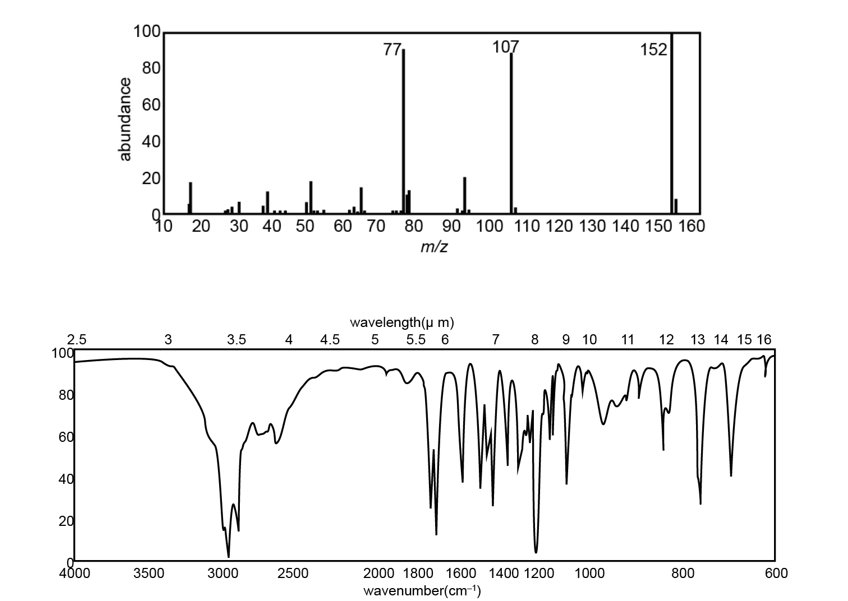
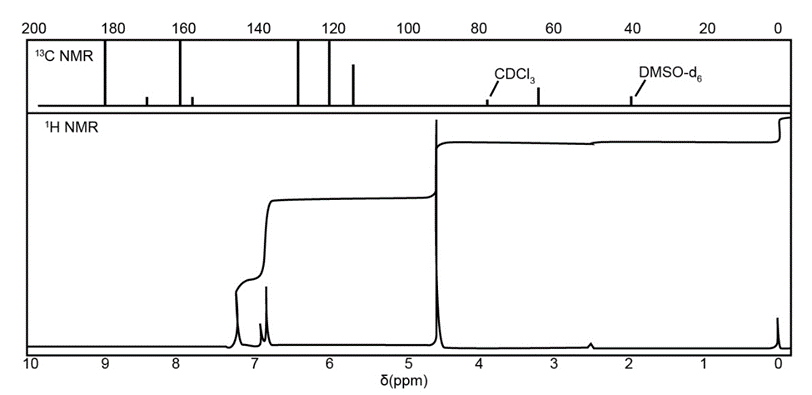
Question:
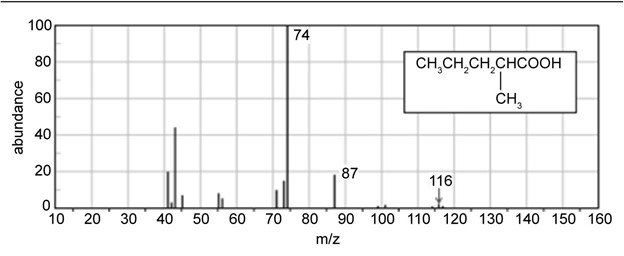
Question: Show how you would synthesize the following carboxylic acids, using the indicated starting materials.
(a)
(b)
(c )
(d)
(e)
(f)
What do you think about this solution?
We value your feedback to improve our textbook solutions.