Chapter 20: Q11P. (page 1057)
Question: Show how you would synthesize the following carboxylic acids, using the indicated starting materials.
(a)
(b)
(c )
(d)
(e)
(f)
Short Answer
(a)
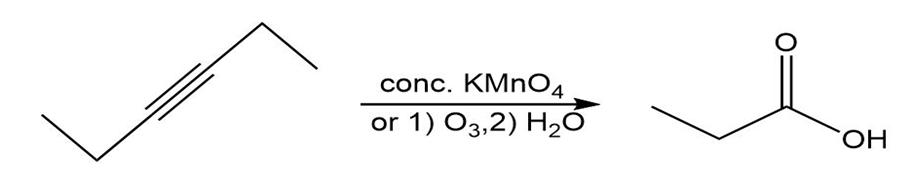
(b)
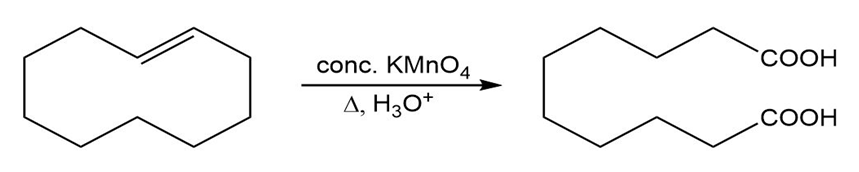
(c)
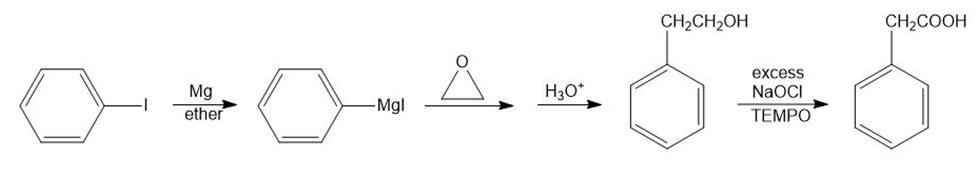
(d)
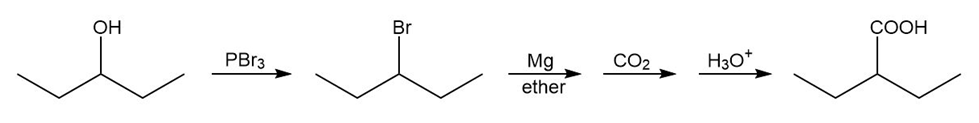
(e)
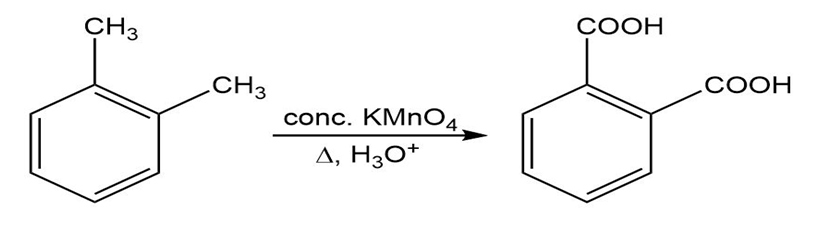
(f)
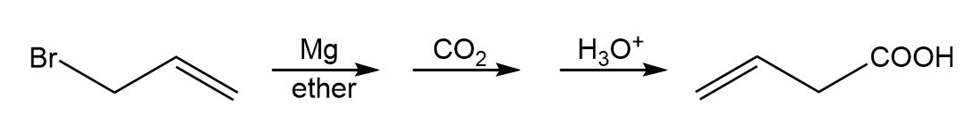
Learning Materials
Features
Discover
Chapter 20: Q11P. (page 1057)
Question: Show how you would synthesize the following carboxylic acids, using the indicated starting materials.
(a)
(b)
(c )
(d)
(e)
(f)
(a)
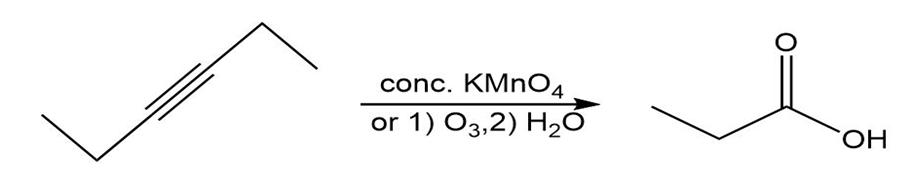
(b)

(c)

(d)

(e)

(f)

All the tools & learning materials you need for study success - in one app.
Get started for free
Question: A carboxylic acid has two oxygen atoms, each with two nonbonding pairs of electrons.
Question: Arrange each group of compounds in order of increasing acidity.
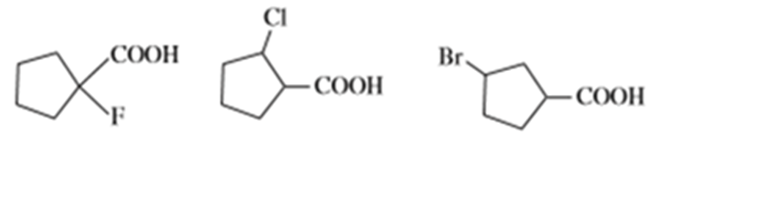
Question:
Show how Fischer esterification might be used to form the following esters. In each case, suggest a method for driving the reaction to completion.
(a)methyl salicylate
(b) methyl formate
(c) ethyl phenylacetate
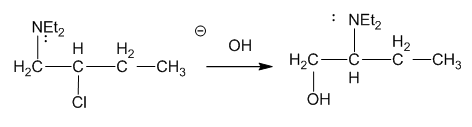
The following reaction takes place under second-order conditions (strong nucleophile), yet the structure of the product shows rearrangement. Also, the rate of this reaction is several thousand times faster than the rate of substitution of hydroxide ion on 2-chlorobutane under similar conditions. Propose a mechanism to explain the enhanced rate and rearrangement observed in this unusual reaction. (“Et” is the abbreviation for ethyl.
What do you think about this solution?
We value your feedback to improve our textbook solutions.