Chapter 20: Q11P (page 1038)
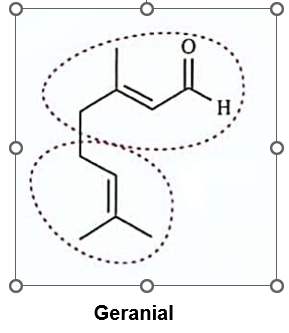
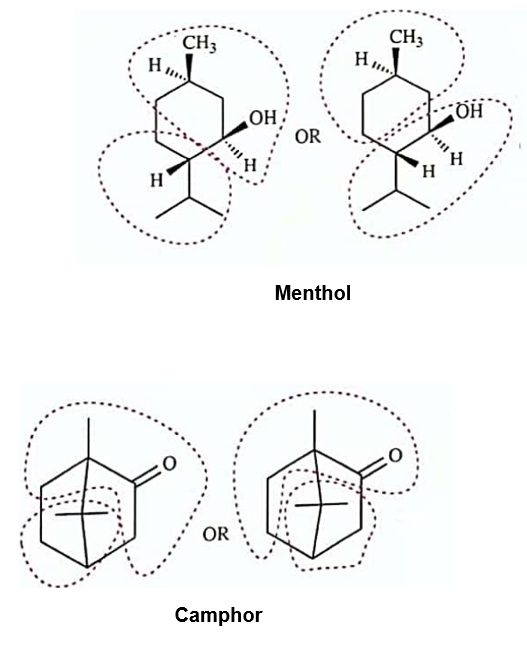
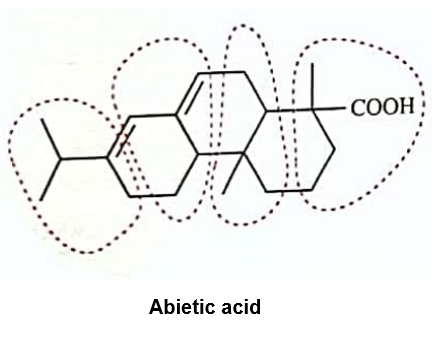
Circle the isoprene units in geranial, menthol, camphor, and abietic acid.
Short Answer
Learning Materials
Features
Discover
Chapter 20: Q11P (page 1038)
Circle the isoprene units in geranial, menthol, camphor, and abietic acid.



All the tools & learning materials you need for study success - in one app.
Get started for free
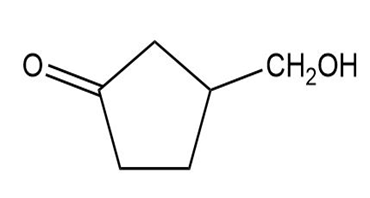
Show how you would synthesize the following compounds from the appropriate carboxylic acids or acid derivatives.
(a)
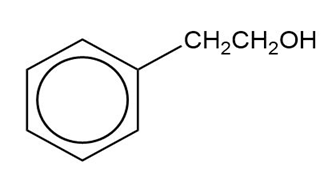
(b)
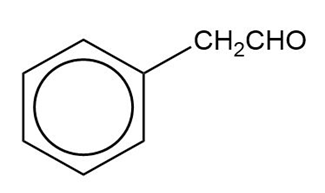
(c)
Dimethylamine(CH3) 2 NHhas a molecular weight of 45 and a boiling point of 7.40C. Trimethylamine, (CH3) 3N, has a higher molecular weight (59) but a lower boiling point(3.50C) . Explain this apparent discrepancy.
Question: Show the products you expect when each compound reacts with NBS with light shining on the reaction.
c)
Question:
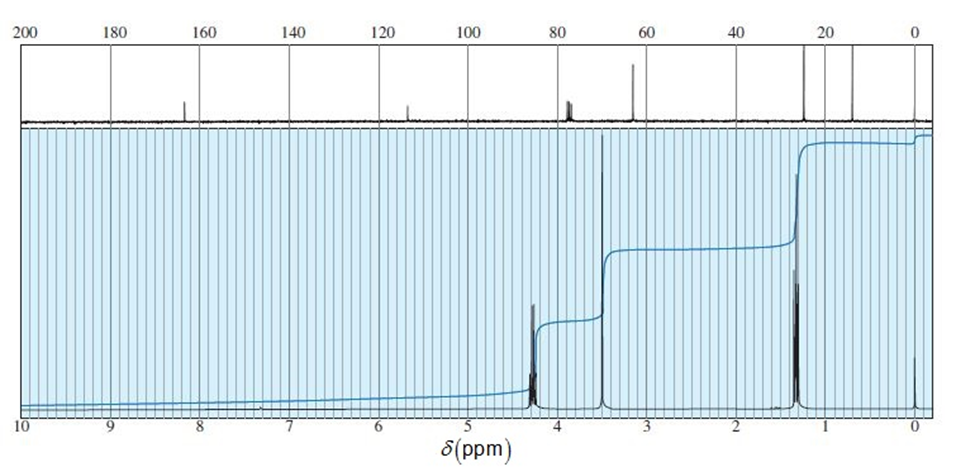
An unknown compound gives a mass spectrum with a weak molecular ion at m/z113 and a prominent ion at m/z68. Its NMR and IR spectra are shown here. Determine the structure, and show how it is consistent with the observed absorptions. Propose a favorable fragmentation to explain the prominent MS peak at m/z68.
What do you think about this solution?
We value your feedback to improve our textbook solutions.