Chapter 20: Q31P (page 1038)
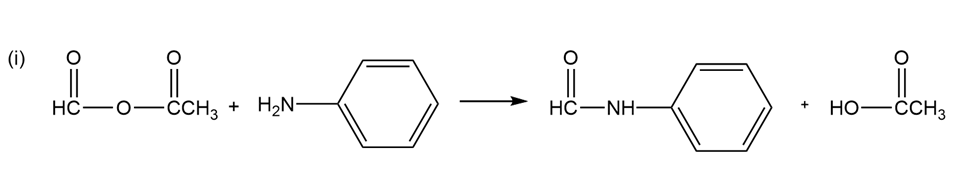
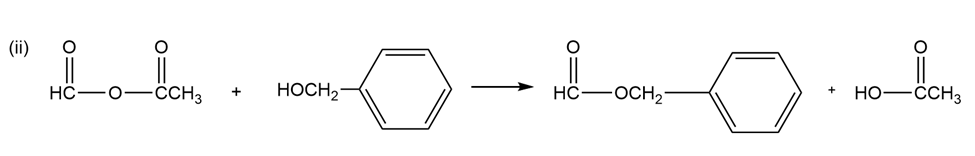
(a) Give the products expected when acetic formic anhydride reacts with
(i) aniline and
(ii) benzyl alcohol
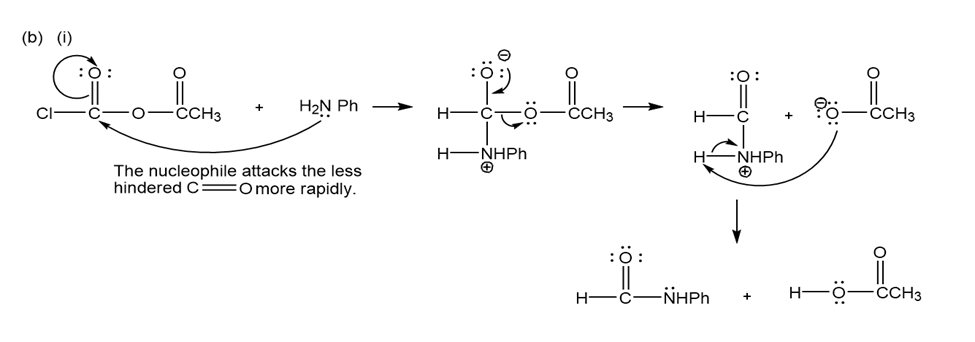
(b) Propose mechanisms for these reactions.
Short Answer
(a)
(b)
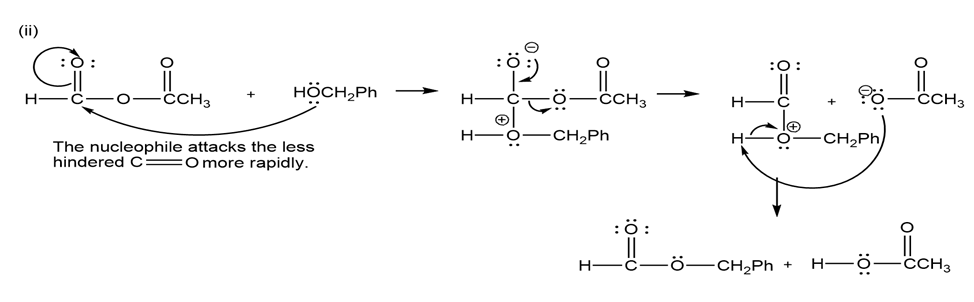
Learning Materials
Features
Discover
Chapter 20: Q31P (page 1038)
(a) Give the products expected when acetic formic anhydride reacts with
(i) aniline and
(ii) benzyl alcohol
(b) Propose mechanisms for these reactions.
(a)


(b)


All the tools & learning materials you need for study success - in one app.
Get started for free
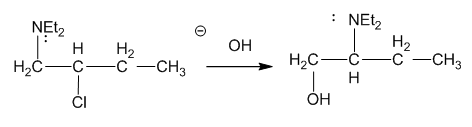
The following reaction takes place under second-order conditions (strong nucleophile), yet the structure of the product shows rearrangement. Also, the rate of this reaction is several thousand times faster than the rate of substitution of hydroxide ion on 2-chlorobutane under similar conditions. Propose a mechanism to explain the enhanced rate and rearrangement observed in this unusual reaction. (“Et” is the abbreviation for ethyl.
Name the following carboxylic acids (when possible, give both a common name and a systematic name).
(a)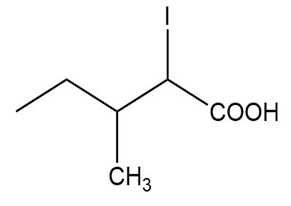
(b)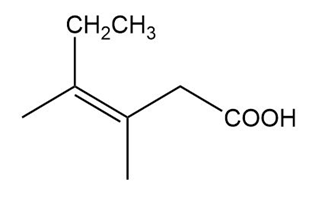
(c)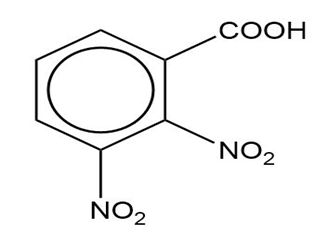
(d)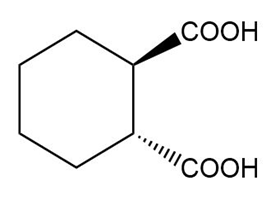
(e)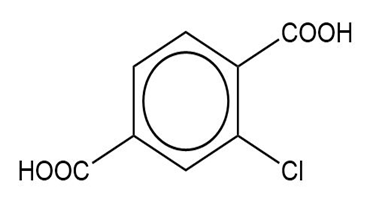
(f)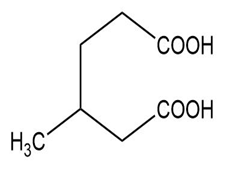
Question: Oxidation of primary alcohol to an aldehyde usually gives some over-oxidation to the carboxylic acid. Assume you have used PCC to oxidize pentan-1-ol to pentanal.
Use chemical equations to show how the following accidents cause injury to the clothing involved (not to mention the skin under the clothing!)
Dimethylamine(CH3) 2 NHhas a molecular weight of 45 and a boiling point of 7.40C. Trimethylamine, (CH3) 3N, has a higher molecular weight (59) but a lower boiling point(3.50C) . Explain this apparent discrepancy.
What do you think about this solution?
We value your feedback to improve our textbook solutions.