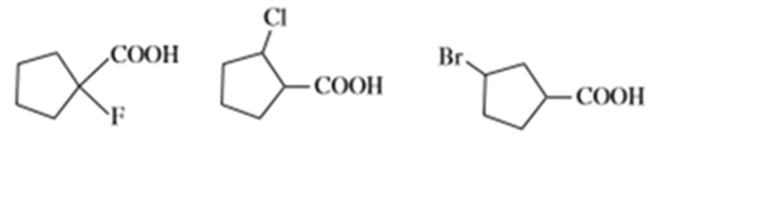
Chapter 20: Q2P. (page 1042)
Question: Name the following carboxylic acids (when possible, give both a common name and a systematic name).
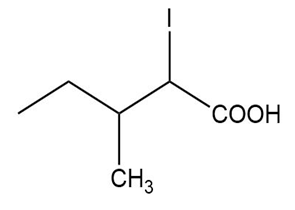
(a)
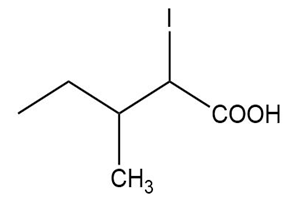
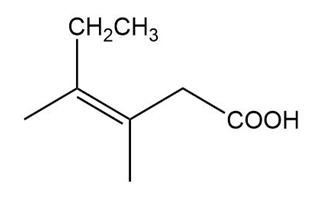
(b)
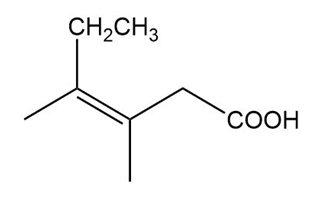
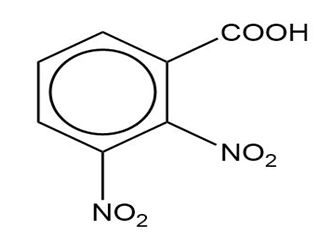
(c)
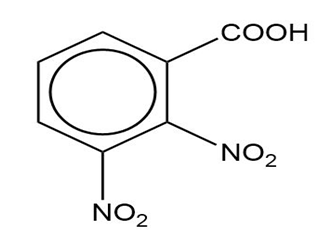
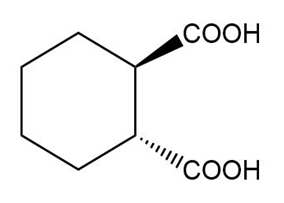
(d)
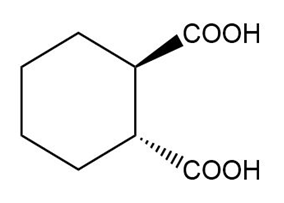
(e)
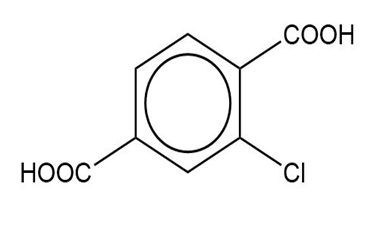
(f)
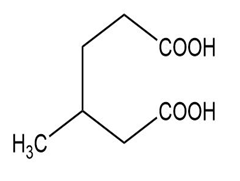
Short Answer
(a) The IUPAC name is 2-iodo-3-methylpentanoic acid, and the common name is
(b) The IUPAC name is (Z)-3,4-dimethylhex-3-enoic acid, and there is no common name.
(c) The IUPAC name is 2,3-dinitrobenzoic acid, and there is no common name.
(d) The IUPAC name is trans-cyclohexane-1,2-dicarboxylic acid, and the common name is trans-hexahydrophthalic acid.
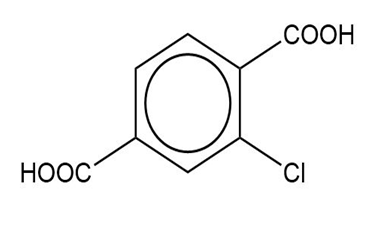
(e) The IUPAC name is 2-chlorobenzene-1,4-dicarboxylic acid, and the common name is 2-chloroterephthalic acid.
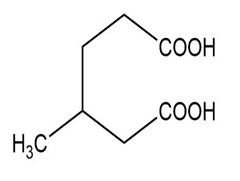
(f) The IUPAC name is 3-methylhexanedioic acid, and the common name is